Abstract
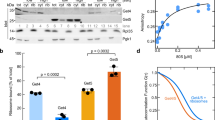
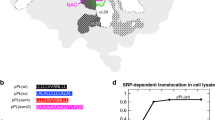
THE signal recognition particle (SRP) consists of one RNA and six protein subunits1,2. The N-terminal domain of the 54K subunit contains a putative GTP-binding site, whereas the C-terminal domain binds signal sequences and SRP RNA3–7. Binding of SRP to the signal sequence as it emerges from the ribosome creates a cytosolic targeting complex containing the nascent polypeptide chain, the translating ribosome, and SRP8. This complex is directed to the endoplasmic reticulum membrane as a result of its interaction with the SRP receptor9–11, a membrane protein composed of two subunits, SRα and SRβ, each of which also contains a GTP-binding domain12,13. In the presence of GTP, SRP receptor binding to SRP causes the latter to dissociate from both the signal sequence and the ribosome13,14. GTP is then hydrolysed so that SRP can be released from the SRP receptor and returned to the cytosol15. Here we show that the 54K subunit (Mr 54,000) of SRP (SRP54) is a GTP-binding protein stabilized in a nucleotide-free state by signal sequences, and that the SRP receptor both increases the affinity of SRP54 for GTP and activates its GTPase. We propose that nucleotide-mediated conformational changes in SRP54 regulate the release of signal sequences and the docking of ribosomes at the endoplasmic reticulum.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Walter, P. & Blobel, G. Proc. natn. Acad. Sci. U.S.A. 77, 7112–7116 (1980).
Walter, P. & Blobel, G. Nature 99, 691–698 (1982).
Bernstein, H. D. et al. Nature 340, 482–486 (1989).
Römisch, K. et al. Nature 340, 478–482 (1989).
Zopf, D., Bernstein, H. D., Johnson, A. E. & Walter, P. EMBO J. 9, 4511–4517 (1990).
Römisch, K., Webb, J., Lingelbach, K., Gausepohl, H. & Dobberstein, B. J. Cell Biol. 111, 1793–1802 (1990).
High, S. & Dobberstein, B. J. Cell Biol. 113, 229–233 (1991).
Nunnari, J. & Walter, P. Curr. Opin. Cell Biol. 4, 573–580 (1992).
Gilmore, R., Blobel, G. & Walter, P. J. Cell Biol. 95, 463–469 (1982).
Gilmore, R., Walter, P. & Blobel, G. J. Cell Biol. 95, 470–477 (1982).
Meyer, D. I., Krause, E. & Dobberstein, B. Nature 297, 647–650 (1982).
Tajima, S., Lauffer, L., Rath, V. L. & Walter, P. J. Cell Biol. 103, 1167–1178 (1986).
Connolly, T. & Gilmore, R. Cell 57, 599–610 (1989).
Gilmore, R. & Blobel, G. Cell 35, 677–685 (1983).
Connolly, T., Rapiejko, P. J. & Gilmore, R. Science 252, 1171–1173 (1991).
Connolly, T. & Gilmore, R. J. Cell. Biol. (in the press).
Walter, P. & Blobel, G. Cell 34, 525–533 (1983).
Siegel, V. & Walter, P. J. Cell Biol. 100, 1913–1921 (1985).
Nath, J. P., Eagle, G. R. & Himes, R. H. Biochemistry 24, 1555–1560 (1985).
Pashev, I. G., Dimitrov, S. I. & Angelov, D. Trends Biochem. Sci. 16, 323–326 (1991).
Bourne, H. R., Sanders, D. A. & McCormick, F. Nature 349, 117–127 (1991).
Emr, S. & Silhavy, T. J. Proc. natn. Acad. Sci. U.S.A. 80, 4599–4603 (1983).
McKnight, C. J., Briggs, M. S. & Gierasch, L. M. J. biol. Chem. 264, 17293–17297 (1989).
Lütcke, H., High, S., Römisch, K., Ashford, A. & Dobberstein, B. EMB0 J. 11, 1543–1551 (1992).
Bourne, H. R., Sanders, D. A. & McCormick, F. Nature 348, 125–132 (1990).
Gilman, A. G. A. Rev. Biochem. 56, 615–649 (1987).
Rapiejko, P. J. & Gilmore, R. J. Cell Biol. 117, 493–503 (1992).
Walter, P. & Blobel, G. Meth. Enzym. 96, 682–691 (1983).
Migliaccio, G., Nicchitta, C. V. & Blobel, G. J. Cell Biol. 117, 15–25 (1992).
Segel, I. H. Enzyme Kinetics: Behavior and Analysis of Steady State and Rapid Equilibrium Enzyme Systems 105 (Wiley-lnterscience, New York, 1975).
Watanabe, M., Hunt, J. F. & Blobel, G. Nature 323, 71–73 (1986).
Chen, L. Tai, P. C., Briggs, M. S. & Gierasch, L. M. J. biol. Chem. 262, 1427–1429 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Miller, J., Wilhelm, H., Gierasch, L. et al. GTP binding and hydrolysis by the signal recognition particle during initiation of protein translocation. Nature 366, 351–354 (1993). https://doi.org/10.1038/366351a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/366351a0
This article is cited by
-
Gene ontology analysis of expanded porcine blastocysts from gilts fed organic or inorganic selenium combined with pyridoxine
BMC Genomics (2018)
-
Alternative 3′ UTRs act as scaffolds to regulate membrane protein localization
Nature (2015)
-
Summing up particular features of protein secretion in Streptomyces lividans
World Journal of Microbiology and Biotechnology (2011)
-
Delivering proteins for export from the cytosol
Nature Reviews Molecular Cell Biology (2009)
-
SRP RNA controls a conformational switch regulating the SRP–SRP receptor interaction
Nature Structural & Molecular Biology (2008)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.