Abstract
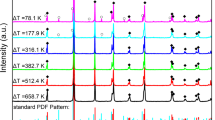
POLYMORPHISM of crystal structures—alternative crystal structures resulting from variations in ionic and molecular packing1 and conformation2—is important in a number of fields in solid-state chemistry, including biomineralization3 (in the mineral phases of calcium carbonate3, for example), polymer science (polypropylene4, for example), explosives (ammonium nitrate, lead azide5), nonlinear optical materials6, ceramics and catalysis (zeolites and metal oxides7). In the pharmaceutical and fine-chemicals industries, polymorphism is central to both production process design and product activity8. Here we show that crystal twinning can stabilize a crystal polymorph that is otherwise not the most stable form. We use electron microscopy, X-ray diffraction and Raman spectroscopy to show that the apparently indefinite persistence of a metastable polymorph of terephthalic acid can be explained on this basis. If twinning can be engineered, it may therefore provide a means for stabilizing crystal phases with useful physical properties.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Verma, A. R. & Krishna, P. Polymorphism and Polytypism in Crystals 33–60 (Wiley, New York, 1966).
Bernstein, J. & Hagler, A. T. J. Am. chem. Soc. 100, 673–681 (1978)
Bischoff, J. L. & Fyfe, W. S. Am. J. Sci. 266, 65–79 (1968).
Keller, A., Goldbeck-Wood, G. & Hikosaka, M. J. chem. Soc., Faraday Disc., No. 95 (in the press).
Hattori, K. & McCrone, W. Analyt. Chem. 28, 1791–1793 (1956).
Hall, S. R. et al. J. Cryst. Growth 79, 745–751 (1986).
Subotic, B. et al. Zeolites 2, 135–142 (1982).
Davey, R. J. in Crystal Growth in Science and Technology NATO ASI Ser. (Eds Arend, H. & Hulliger, J.) 217–224 (Plenum, New York, 1989).
Bailey, M. & Brown, C. J. Acta crystallogr. 22, 387–391 (1967).
Berkovitch-Yellin, Z. & Leiserowitz, L. J. Am. chem. Soc. 104, 4052–4064 (1982).
Gerasirnov, V. P., Zharikov, N. K., Korobkov, V. S., Ovchinnikov, I. V. & Rud, L. V. Zh. Prikl. Spectroskopii 34, 308–311 (1981).
Saska, M. & Myerson, A. S. Cryst. Res. Technol. 20, 201–208 (1985).
Bhat, H. L. Clarke, S. M., El Korashy, A., & Roberts, K. J. J. appl. Crystallogr. 23, 545–549 (1990).
Ozaki, T. & Shigeyasu, M. Maruzen Sekiyu Giho 20, 81–90 (1975).
Ostwald, W. Z. phys. Chem. 22, 289–330 (1897).
Avrami, M. J. chem. Phys. 7, 1103–1112 (1939).
Cardew, P. T., Davey, R. J. & Ruddick, A. J. J. chem. Soc. Faraday Trans. 80, 659–668 (1984).
Mnjukh, Ju. V. Molec. Cryst. Liq. Cryst. 52, 201–218 (1979).
Davey, R. J. et al. J. Phys. D 24, 176–185 (1991).
Davey, R. J. et al. J. chem. Soc., Faraday Trans. 88, 3461–3466 (1992).
Davey, R. J., Harding, M. M. & Rule, R. J. J. Cryst. Growth. 144, 7–12 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Davey, R., Maginn, S., Andrews, S. et al. Stabilization of a metastable crystalline phase by twinning. Nature 366, 248–250 (1993). https://doi.org/10.1038/366248a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/366248a0
This article is cited by
-
Preparation of LTA, HS and FAU/EMT intergrowth zeolites from aluminum scraps and industrial metasilicate
Journal of Material Cycles and Waste Management (2019)
-
Incompatible Sets of Gradients and Metastability
Archive for Rational Mechanics and Analysis (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.