Abstract
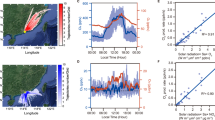
SPRINGTIME ozone depletion over Antarctica is thought1,2 to be due to catalytic cycles involving chlorine monoxide, which is formed as a result of reactions on the surface of polar stratospheric clouds (PSCs). When the PSCs evaporate, CIO in the polar air can react with NO2 to form the reservoir species C1ONO2. High concentrations of C1ONO2 can also be found at lower latitudes because of direct transport of polar air or mixing of CIO and NO2 at the edges of the polar vortex. C1ONO2 can take part in an ozone-depleting catalytic cycle18, but the significance of this cycle has not been clear. Here we present model simulations of ozone concentrations from March to May both within the Arctic vortex and at a mid-latitude Northern Hemisphere site. We find increasing ozone loss from March to May. The C1ONO2 cycle seems to be responsible for a significant proportion of the simulated ozone loss. An important aspect of this cycle is that it is not as limited as the other chlorine cycles to the timing and location of PSCs; it may therefore play an important role in ozone depletion at warm middle latitudes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Report No. 25 (World Meteorological Organisation, Geneva, 1992).
Solomon, S. Nature 347, 347–354 (1990).
Toon, G. C. et al. J. geophys. Res. 94, 16571–16596 (1989).
Toon, G. C., Farmer, C. B., Shaper, P. W., Lowes, L. L. & Norton, R. H. J. geophys. Res. 97, 7939–7961 (1992).
Oelhaf, H. et al. Geophys. Res. Lett. (in the press).
Toumi, R., Bekki, S. & Cox, R. J. atmos. Chem. 16, 135–144 (1993).
Austin, J., Butchart, N. & Shine, K. P. Nature 360, 221–225 (1992).
Yung, Y. L., Pinto, J. P., Watson, R. T. & Sander, S. P. J. atmos. Sci. 37, 339–353 (1980).
Margitan, J. J. J. phys. Chem. 87, 674–679 (1983).
Minton, T. K., Nelson, C. M., Moore, T. A. & Okumara, M. Science 258, 1342–1345 (1992).
Wayne, R. P. (ed.) Atmos. Envir. 25A, no. 1, special issue (1991).
Molina, L. T. & Molina, M. J. J. phys. Chem. 91, 433–436 (1987).
Fahey, D. W. et al. Nature 363, 509–514 (1993).
Zander, R. et al. J. atmos. Chem. 15, 171–186 (1992).
Molina, M. & Rowland, F. S. Nature 249, 810–814 (1974).
Solomon, S., Garcia, R. R., Rowland, F. S. & Wuebbles, D. J. Nature 321, 755–758 (1986).
DeMore, et al. Eval. No. 10, JLP-92 (NASA Jet Propulsion Lab., Pasadena, 1992).
Chang, J. S., Barker, R. R., Davenport, J. E. & Goldan, D. M. Chem. Phys. Lett. 60, 385–390 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Toumi, R., Jones, R. & Pyle, J. Stratospheric ozone depletion by CIONO2 photolysis. Nature 365, 37–39 (1993). https://doi.org/10.1038/365037a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/365037a0
This article is cited by
-
The potential impact of the reaction OH+ClO?HCl+O2 on polar ozone photochemistry
Journal of Atmospheric Chemistry (1995)
-
Sensitivity of stratospheric composition to oxygen absorption of solar radiation (175?210 nm)
Journal of Atmospheric Chemistry (1994)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.