Abstract
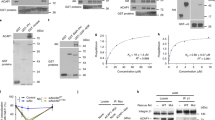
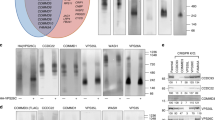
THE formation of constitutive transport vesicles involves the association of non-clathrin coat proteins to transport organelles1,2. Here we report that IgE receptors and protein kinase C (PKC) regulate the GTP-dependent binding of the two coat proteins ADP-ribosylation factor (ARF) and β-COP3–5 to Golgi membranes in rat basophilic leukaemia cells. Activation of IgE receptors and PKC prevented the ARF and β-COP dissociation from Golgi membranes that occurs in permeabilized cells in the absence of GTP and potentiated the association-promoting effects of GTP and the G protein activator fluoroluminate. In contrast, PKC downregulation and PKC inhibition abolished the activity of GTP and fluoroluminate in promoting ARF binding to the Golgi complex. Studies of ARF binding to isolated Golgi membranes gave similar results. These findings suggest that coat assembly on Golgi membranes, and thus possibly constitutive secretory traffic, is modulated by membrane receptors and second messengers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Rothman, J. E. & Orci, L. Nature 355, 409–415 (1992).
Melangon, P., Franzusoff, A. & Howell, K. E. Trends Cell Biol. 1, 165–171 (1991).
Kahn, R. A. et al. J. biol. Chem. 267, 13039–13046 (1992).
Serafini, T. et al. Cell 67, 239–253 (1991).
Balch, W. E., Kahn, R. A. & Schwaninger, R. J. biol. Chem. 267, 13053–13061 (1992).
Stearns, T., Willingham, M. C., Botstein, D. & Kahn, R. A. Proc. natn. Acad. Sci. U.S.A. 87, 1238–1242 (1990).
Allan, V. & Kreis, T. E. J. Cell Biol. 103, 2229–2239 (1986).
Donaldson, J. G., Kahn, R. A., Lippincott-Schwartz, J. & Klausner, R. D. Science 254, 1197–1199 (1991).
Orci, L. et al. Cell 64, 1183–1195 (1991).
De Matteis, M. A., Di Tullio, G., Buccione, R. & Luini, A. J. biol. Chem. 266, 10452–10460 (1991).
Kahn, R. A. J. biol. Chem. 266, 15595–15597 (1991).
Donaldson, J. G., Cassel, D., Kahn, R. A. & Klausner, R. D. Proc. natn. Acad. Sci. U.S.A. 89, 6408–6412 (1992).
Stow, J. L. et al. J. Cell Biol. 114, 1113–1124 (1991).
Ktistakis, N. T., Linder, M. E. & Roth, M. G. Nature 356, 344–346 (1992).
Ali, H., Collado-Escobar, D. M. & Beaven, M. A. J. Immun. 143, 2626–2633 (1989).
Kawakami, T. et al. J. Immun. 148, 3513–3519 (1992).
Huang, F. L., Yoshida, Y., Cunha-Melo, J. R., Beaven, M. A. & Huang, K.-P. J. biol. Chem. 264, 4238–4243 (1989).
Bruns, R. F. et al. Biochem. biophys. Res. Commun. 176, 288–293 (1991).
Azzi, A., Boscoboinik, D. & Hensey, C. Eur. J. Biochem. 208, 547–557 (1992).
Donaldson, J. G., Finazzi, D. & Klausner, R. D. Nature 360, 350–352 (1992).
Helms, J. B. & Rothman, J. E. Nature 360, 352–354 (1992).
Ozawa, K. et al. J. biol. Chem. 268, 1749–1756 (1993).
Klausner, R. D., Donaldson, J. G. & Lippincott-Schwartz, J. J. Cell Biol. 116, 1071–1080 (1992).
Pelham, H. R. B. Cell 67, 449–451 (1991).
Hosobuchi, M., Kreis, T. & Schekman, R. Nature 360, 603–605 (1992).
Miller, S. G. & Moore, H.-P. H. J. Cell Biol. 112, 39–54 (1991).
Burgess, T. L. & Kelly, R. B. A. Rev. Cell Biol. 3, 243–292 (1987).
Cunha-Melo, J. R. et al. J. Immun. 143, 2617–2625 (1989).
Strous, G. J., van Kerhof, P., van Meer, G., Rijnboutt, S. & Stoorvogel, W. J. biol. Chem. 268, 2341–2347 (1993).
Miller, S. G., Carnell, L. & Moore, H.-P. H. J. Cell Biol. 118, 267–283 (1992).
Barr, F. A. et al. FEBS Lett. 294, 239–243 (1991).
Arvan, P. & Castle, D. Trends Cell Biol. 2, 327–331 (1992).
Balch, W. E., Dunphy, W. G., Braell, W. A. & Rothman, J. E. Cell 39, 405–416 (1984).
Malhotra, V., Serafini, T., Orci, L., Shepherd, J. C. & Rothman, J. E. Cell 58, 329–336 (1989).
Low, S. H. et al. J. biol. Chem. 266, 17729–17732 (1991).
Hunziker, W., Whitney, A. & Mellman, I. Cell 67, 617–627 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
De Matteis, M., Santini, G., Kahn, R. et al. Receptor and protein kinase C-mediated regulation of ARF binding to the Golgi complex. Nature 364, 818–821 (1993). https://doi.org/10.1038/364818a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/364818a0
This article is cited by
-
Regulation of Golgi signaling and trafficking by the KDEL receptor
Histochemistry and Cell Biology (2013)
-
A traffic-activated Golgi-based signalling circuit coordinates the secretory pathway
Nature Cell Biology (2008)
-
The physiology of membrane transport and endomembrane-based signalling
The EMBO Journal (2006)
-
Differential roles of Ras and Rap1 in growth factor-dependent activation of phospholipase Cε
Oncogene (2002)
-
Adaptor proteins in protein kinase C-mediated signal transduction
Oncogene (2001)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.