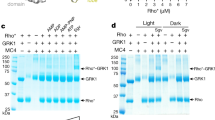
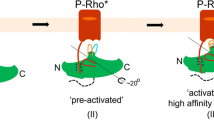
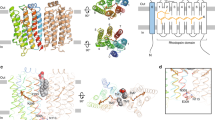
Abstract
RHODOPSIN kinase1 and β-adrenergic receptor kinase (β ARK)2 are related members of a serine/threonine kinase family that specifically initiate deactivation of G-protein-coupled receptors. After stimulus-mediated receptor activation, these cytoplasmic kinases translocate to the plasma membrane3,4. Here we show that the molecular basis for this event involves a class of unsaturated lipids called isoprenoids. Covalent modification in vivo of rhodopsin kinase by a 15-C (farnesyl) isoprenoid5 enables the kinase to anchor to photon-activated rhodopsin. Mutations that alter or eliminate the isoprenoid, fully disable light-specific Rhodopsin kinase translocation. Other receptor kinases (such as βARK), which lack an intrinsic lipid, are activated6 on exposure to brain βγ subunits of the signal-transducing G proteins, the γ subunit of which bears a 20-C (geranylgeranyl) isoprenoid7,8. Using chimaeric β ARKs that undergo isoprenylation in vitro, we demonstrate that membrane association and activation of these kinases can occur in the absence of βγ. These results indicate that rhodopsin kinase (by means of an integral isoprenoid) and β ARK (through its association with βγ) both rely on the function of isoprenyl moieties for their translocation and activity, illustrating distinct, though related, modes of biological regulation of receptor function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lorenz, W. et al. Proc. natn. Acad. Sci. U.S.A. 88, 8715–8719 (1991).
Benovic, J. L., DeBlasi, A., Stone, W. C., Caron, M. G. & Lefkowitz, R. J. Science 246, 235–240 (1989).
Kuhn, H. Biochemistry 21, 4389–4395 (1978).
Strasser, R. H., Benovic, J. L., Caron, M. G. & Lefkowitz, R. J. Proc. natn. Acad. Sci. U.S.A. 83, 6362–6366 (1986).
Inglese, J., Glickman, J. F., Lorenz, W., Caron, M. G. & Lefkowitz, R. J. J. biol. Chem. 267, 1422–1425 (1992).
Haga, K. & Haga, T. J. biol. Chem. 267, 2222–2227 (1992).
Mumby, S. M., Casey, P. J., Gilman, A. G., Gutowski, S. & Sternweis, P. C. Proc. natn. Acad. Sci. U.S.A. 87, 5873–5877 (1990).
Yamane, H. K. et al. Proc. natn. Acad. Sci. U.S.A. 87, 5868–5872 (1990).
Goldstein, J. L. & Brown, M. S. Nature 343, 425–430 (1990).
Glomset, J. A., Gelb, M. H. & Farnsworth, C. C. Trends biochem. Sci. 15, 139–142 (1990).
Hancock, J. F., Magee, A. I., Childs, J. E. & Marshall, C. J. Cell 57, 1167–1177 (1989).
Willumsen, B. M., Norris, K., Papageorge, A. G., Hubbert, N. L. & Lowy, D. R. EMBO J. 3, 2581–2585 (1984).
Jackson, J. H. et al. Proc. natn. Acad. Sci. U.S.A. 87, 3042–3046 (1990).
Casey, P. J., Solski, P. A., Der, D. C. & Buss, J. E. Proc. natn. Acad. Sci. U.S.A. 86, 8323–8327 (1989).
Cox, A. D. & Der, C. J. Crit. Rev. Oncogen. (in the press).
Pitcher, J. et al. Science (in the press).
Mayor, F., Benovic, J. L., Caron, M. G. & Lefkowitz, R. J. J. biol. Chem. 262, 6468–6471 (1987).
Benovic, J. L., Mayor, F., Somers, R. L., Caron, M. G. & Lefkowitz, R. J. Nature 322, 869–872 (1986).
Simonds, W. F., Butrynski, J. E., Gautam, N., Unson, C. G. & Spiegel, A. M. J. biol. Chem. 266, 5363–5366 (1991).
Simon, M. I., Strathmann, M. P. & Gautam, N. Science 252, 802–808 (1991).
Casey, P. J., Thissen, J. A. & Moomaw, J. F. Proc. natn. Acad. Sci. U.S.A. 88, 8631–8635 (1991).
Kinsella, B. T., Erdman, R. A. & Maltese, W. A. Proc. natn. Acad. Sci. U.S.A. 88, 8934–8938 (1991).
Hancock, J. F., Cadwallander, K., Paterson, H. & Marshall, C. J. EMBO J. 10, 4033–4039 (1991).
Andersson, S., Davis, D. L., Dahlback, H., Jornvall, H. & Russell, D. W. J. biol. Chem. 264, 8222–8229 (1989).
Farnsworth, C. C., Casey, P. J., Howald, W. N., Glomset, J. A. & Gelb, M. H. Methods 1, 231–240 (1990).
Arriza, J. L. et al. J. Neurosci. (in the press).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Inglese, J., Koch, W., Caron, M. et al. Isoprenylation in regulation of signal transduction by G-protein-coupled receptor kinases. Nature 359, 147–150 (1992). https://doi.org/10.1038/359147a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/359147a0
This article is cited by
-
Mechanistic diversity involved in the desensitization of G protein-coupled receptors
Archives of Pharmacal Research (2021)
-
G protein-coupled receptor kinases as therapeutic targets in the heart
Nature Reviews Cardiology (2019)
-
Multiple functions of G protein-coupled receptor kinases
Journal of Molecular Signaling (2014)
-
Protein Prenylation: An (Almost) Comprehensive Overview on Discovery History, Enzymology, and Significance in Physiology and Disease
Monatshefte für Chemie - Chemical Monthly (2006)
-
Interaction between simvastatin and metoprolol with respect to cardiac β-adrenoceptor density, catecholamine levels and perioperative catecholamine requirements in cardiac surgery patients
Naunyn-Schmiedeberg's Archives of Pharmacology (2005)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.