Abstract
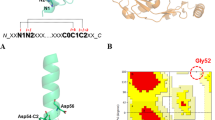
THE rational design of proteins requires knowledge of the helix-forming propensities (s-values) of the different amino acids. There is, however, considerable controversy about the relative values for alanine and glycine1–12We find from experiments on mutants of barnase that the relative effect of Ala versus Gly on helix stability depends crucially on the position in the helix (whether they are at the ends (caps) or are internal) and the context (the influence of their neighbours). Glycine is greatly preferred at the N and C caps. At internal positions, Ala stabilizes the helix relative to Gly by 0.4 to 2 kcal mol−1. The variation results from a combination of burial of hydrophobic surface on folding and interference with hydrogen bonding of the protein with solvent. There is a good empirical correlation between the relative stabilizing effects of Ala and Gly with the total change in solvent-accessible hydrophobic surface area of the folded protein on mutation of Gly to Ala. It is not valid to assign to each amino acid a unique s-value that is generally applicable to all positions in all helices in all proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Sueki et al. Macromolecules 17, 148–155 (1984).
Scheraga, H. A. Proc. natn. Acad. Sci U.S.A. 82, 5585–5587 (1985).
Altmann, K. H., Wojcik, J., Vasquez, M. & Scheraga, H. A. Biopolymers 30, 107–120 (1990).
Padmanabhan, S., Marqusee, S., Ridgeway, T., Laue, T. M. & Baldwin, R. L. Nature 344, 268–270 (1990).
Merutka, G., Lipton, W., Shalongo, W., Park, S. H. & Stellwagen, E. Biochemistry 29, 7511–7515 (1990).
Lyu, P. C., Lip, M. I., Marky, L. A. & Kallenbach, N. R. Science 250, 669–673 (1990).
Strehlow, K. G. & Baldwin, R. L. Biochemistry 28, 2130–2133 (1989).
O'Neil, K. T. & DeGrado, W. F. Science 250, 646–651 (1990).
Marqusee, S., Robbins, V. H. & Baldwin, R. L. Proc. natn. Acad. Sci. U.S.A. 86, 5286–5290 (1989).
Chakrabartty, A., Schellman, J. A. & Baldwin, R. L. Nature 351, 586–588 (1991).
Kemp, D. S., Boyd, J. G. & Muendel, C. C. Nature 352, 451–454.
Lesk, A. M. Nature 352, 379–380.
Altman, K. H., Wojcik, J., Vasques, M. & Scheraga, H. A. Biopolymers 30, 107–120 (1990).
Lifson, S. & Roig, A. J. J. chem. Phys. 34, 1963–1974 (1961).
Serrano, L. & Fersht, A.R. Nature 342, 296–299 (1989).
Sali, D., Bycroft, M. & Fersht, A. R. Nature 335, 563–567 (1988).
Kellis, J. T. Jr, Nyberg, K., Sali, D. & Fersht, A. R. Nature 333, 784–786 (1988).
Kellis, J. T. Jr, Nyberg, K. & Fersht, A. R. Biochemistry 28, 4914–4922 (1989).
Horovitz, A., Serrano, L., Avron, B., Bycroft, M. & Fersht, A. R. J. molec. Biol. 216, 1031–1044 (1990).
Horovitz, A., Serrano, L. & Fersht, A. R. J. molec. Biol. 219, 5–9 (1991).
Serrano, L., Bycroft, M. & Fersht, A. R. J. molec. Biol. 218, 465–475 (1991).
Presta, L. G. & Rose, G. D. Science 240, 2632–1641 (1988).
Richardson, J. S. & Richardson, D. C. Science 240, 1648–1652 (1988).
Hermann, R. B. J. phys. Chem. 76, 2754–2759 (1972).
Harris, M. J., Higuchi, T. & Rytting, J. J. phys. Chem. 77, 2694–2702 (1972).
Reynolds, J. A., Gilbert, D. B. & Tanford, C. Proc. natn. Acad. Sci. U.S.A. 71, 2925–2927 (1974).
Chothia, C. Nature 248, 338–339 (1974).
Kellis, J. T. Jr, Nyberg, K. & Fersht, A. R. Biochemistry 28, 4914–4922 (1989).
Serrano, L., Kellis, J. T., Cann, P., Matouschek, A. & Fersht, A. R. J. molec. Biol. (in the press).
Sharp, K. A., Nicholls, A., Friedman, R. & Honig, B. Biochemistry 30, 9686–9697 (1991).
Matthews, B. W., Nicholson, H. & Becktel, W. J. Proc. natn. Acad. Sci. U.S.A. 84, 6663–6667 (1987).
Richards, F. M. & Richmond, T. Ciba Fdn Symp. 60, 23–37 (1978).
Shrake, A. & Rupley, J. A. J. molec. Biol. 79, 351–371 (1973).
Miller, S., Janin, J., Lesk, A. M. & Chothia, C. J. molec. Biol. 196, 641–656 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Serrano, L., Neira, JL., Sancho, J. et al. Effect of alanine versus glycine in α-helices on protein stability. Nature 356, 453–455 (1992). https://doi.org/10.1038/356453a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/356453a0
This article is cited by
-
Directed evolution of a genetically encoded immobilized lipase for the efficient production of biodiesel from waste cooking oil
Biotechnology for Biofuels (2019)
-
Dissecting genetic factors affecting phenylephrine infusion rates during anesthesia: a genome-wide association study employing EHR data
BMC Medicine (2019)
-
Nanoplastics can change the secondary structure of proteins
Scientific Reports (2019)
-
Protein Evolution is Potentially Governed by Protein Stability: Directed Evolution of an Esterase from the Hyperthermophilic Archaeon Sulfolobus tokodaii
Journal of Molecular Evolution (2018)
-
Structure of a LOV protein in apo-state and implications for construction of LOV-based optical tools
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.