Abstract
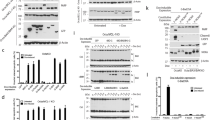
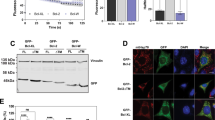
Following exposure of cells to stimuli that trigger programmed cell death (apoptosis), cytochrome c is rapidly released from mitochondria into the cytoplasm where it activates proteolytic molecules known as caspases that specifically cleave the amino-acid sequence DEVD and are crucial for the execution of apoptosis1,2,3,4. The protein Bcl-2 interferes with this activation of caspases by preventing the release of cytochrome c2,3,4. Here we study these molecular interactions during apoptosis induced by the protein Bax, a pro-apoptotic homologue of Bcl-2 (refs 5, 6). We show that in cells transiently transfected with bax, Bax localizes to mitochondria and induces the release of cytochrome c, activation of caspase-3, membrane blebbing, nuclear fragmentation, and cell death. Caspase inibitors do not affect Bax-induced cytochrome c release but block caspase-3 activation and nuclear fragmentation. Unexpectedly, Bcl-2 also fails to prevent Bax-induced cytochrome c release, although it co-localizes with Bax to mitochondria. Cells overexpressing both Bcl-2 and Bax show no signs of caspase activation and survive with significant amounts of cytochrome c in the cytoplasm. These findings indicate that Bcl-2 can interfere with Bax killing downstream of and independently of cytochrome c release.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Liu, X., Kim, C. N., Yang, J., Jemmerson, R. & Wang, X. Induction of apoptotic program in cell-free extracts: requirement for dATP and cytochrome c. Cell 86, 147–157 (1996).
Kluck, R. M., Bossy-Wetzel, E., Green, D. R. & Newmeyer, D. D. The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science 275, 1132–1136 (1997).
Yang, J.et al. Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science 275, 1129–1132 (1997).
Kluck, R. M.et al. Cytochrome c activation of CPP32-like proteolysis plays a critical role in a Xenopus cell-free apoptosis system. EMBO J. 16, 4639–4649 (1997).
Oltvai, Z. N., Milliman, C. L. & Korsmeyer, S. J. Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programmed cell death. Cell 74, 609–619 (1993).
Yin, X.-M., Oltvai, Z. N. & Korsmeyer, S. J. BH1 and BH2 domains of Bcl-2 are required for inhibition of apoptosis and heterodimerization with Bax. Nature 369, 321–323 (1994).
Chittenden, T.et al. Aconserved domain in Bak, distinct from BH1 and BH2, mediates cell death and protein binding functions. EMBO J. 14, 5589–5596 (1995).
Otter, I.et al. The binding properties and biological activities of Bcl-2 and Bax in cells exposed to apoptotic stimuli. J. Biol. Chem. (in the press).
Xiang, J., Chao, D. T. & Korsmeyer, S. J. Bax-induced cell death may not require interleukin-1β-converting enzyme-like proteases. Proc. Natl Acad. Sci. USA 93, 14559–14563 (1996).
McCarthy, N. J., Whyte, M. K. B., Gilbert, C. S. & Evan, G. I. Inhibition of Ced-3/ICE-related proteases does not prevent cell death induced by oncogenes, DNA damage, or the Bcl-2 homologue Bak. J. Cell Biol. 136, 215–227 (1997).
Zou, H., Henzel, W. J., Liu, X., Lutschg, A. & Wang, X. Apaf-1, a human protein homologous to C. elegans CED-4, participates in cytochrome c-dependent activation of caspase-3. Cell 90, 405–413 (1997).
Borner, C.et al. The protein bcl-2α does not require membrane attachment, but two conserved domains to suppress apoptosis. J. Cell Biol. 126, 1059–1068 (1994).
Nicholson, D. W.et al. Identification and inhibition of the ICE/CED-3 protease necessary for mammalian apoptosis. Nature 376, 37–43 (1995).
Acknowledgements
We thank D. Nicholson for the anti-caspase-3 antibody, and S. Rusconi, J.-L. Dreyer and M. Wymann for critically reading the manuscript. This research was supported by the Swiss National Science Foundation, the Swiss Cancer League and the Foundation for Aging Research (AETAS). B.J. was supported by the Austrian Science Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rossé, T., Olivier, R., Monney, L. et al. Bcl-2 prolongs cell survival after Bax-induced release of cytochrome c. Nature 391, 496–499 (1998). https://doi.org/10.1038/35160
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35160
This article is cited by
-
Management of isocitrate dehydrogenase 1/2 mutated acute myeloid leukemia
Leukemia (2024)
-
Effects of different remote ischemia perconditioning methods on cerebral infarct volume and neurological impairment in rats
Scientific Reports (2023)
-
Phototoxicity-free blue light for enhancing therapeutic angiogenic efficacy of stem cells
Cell Biology and Toxicology (2023)
-
Recent advances in organic and polymeric carriers for local tumor chemo-immunotherapy
Science China Technological Sciences (2022)
-
Repeated controlled ovarian stimulation-induced ovarian and uterine damage in mice through the PI3K/AKT signaling pathway
Human Cell (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.