Abstract
Cholera is a severe diarrhoeal disease triggered by a toxin produced by specific biotypes of the bacterium Vibrio cholerae that are pathogenic only to humans. There seems to be no chronic state of the disease and the natural reservoir of the pathogen is environmental1. Here we show that egg masses of the non-biting midge Chironomus sp. (Diptera) harbour V. cholerae and act as its sole carbon source, thereby providing a possible natural reservoir for the cholera bacterium.
Similar content being viewed by others
Main
Chironomids are the most widely distributed and often the most abundant insect in fresh water2. Females deposit egg masses, each containing hundreds of eggs encased in a layer of gelatin, at the water's edge — a convenient location for bacteria to exploit this nutrient-rich substrate.
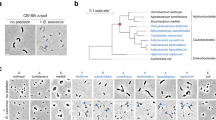
Two hundred floating Chironomus egg masses (Fig. 1a, b) collected from a waste-stabilization pond settled out overnight as thousands of individual eggs (Fig. 1c), most of which did not hatch. To test whether bacteria feeding on and destroying the gelatin matrix might account for this phenom enon, we extracted freshly collected egg masses with a minimal salt-solution medium containing no carbon source3 in order to isolate and identify any bacteria present.
The eggs are arranged in a row, folded into loops to form a spiral, and embedded in a thick, gelatinous cylinder. Egg masses of Chironomus spp. are found in freshwater habitats, where they can reach 25 × 5 mm in size and contain about 1,000 eggs. The appearance of several thousand egg masses at one site is not unusual and, in extreme cases, gelatinous layers several centimetres thick are visible from a distance10,11. a, String-shaped mass of Chironomus luridus eggs (original magnification, × 4). b, Enlarged portion of the mass, showing the gelatinous matrix. c, Enlarged portion 5–10 h after exposure to V. cholerae. Eggs are no longer arranged in spiral rows but protrude from the gelatin, which has been partially consumed by the bacteria. Original magnification in b and c, × 40.
Four isolates were selected and identified as Vibrio cholerae non-O1 non-O139 by standard microbiological tests, as well as by their serotype and fatty-acid profiles. We then collected Chironomus egg masses from other waste-stabilization ponds in various regions of Israel and were able to isolate V. cholerae from all of these samples.
We inoculated fresh egg masses in a salt-solution medium with 1 × 103 V. cholerae per ml. Two controls were run concurrently: egg masses incubated alone under the same conditions, and medium without egg masses inoculated with 1 × 103 per ml V. cholerae. We determined growth of V. cholerae after 24 h by plating samples on thiosulphate–citrate–bile salt substrate.
The number of bacterial colony-forming units (CFUs) that developed in the first control was always less than 0.1% of that recovered in the treated samples; CFUs recovered in the second control did not change. In the medium containing egg masses as the sole carbon source, V. cholerae reached 2 × 106 CFU ml−1. Similar results were obtained when sterilized egg masses were provided as a carbon source. These findings show that egg masses can provide a carbon source to support the development and multiplication of V. cholerae.
Although the V. cholerae biotypes isolated here are non-pathogenic, it is likely that chironomid egg masses would also be a suitable (and abundant) substrate for the pathogenic V. cholerae O1 and O139, assuming that the microhabitat of the pathogenic biotypes is similar4.
An association has been noted between both viable and 'viable but non-culturable' V. cholerae and zooplankton, and copepods have been implicated in the spread of cholera5,6. Propagules may be carried by marine zooplankton along the continental seashore, aided by climatic events such as the El Niño Southern Oscillation7,8,9. These results are relevant to the dispersion of pandemics and to the autochthonous existence of V. cholerae in endemic locales during periods between epidemics — when there is local build-up of the bacterium but no outbreak of disease. Our findings indicate that chironomid egg masses may serve as an intermediate 'host' reservoir for V. cholerae, facilitating its survival and multiplication in freshwater bodies.
References
Heidelberg, F. J. et al. . Nature 406, 477–484 (2000).
Armitage, P., Cranston, P. S. & Pinder, L. C. V. (eds) The Chironomidae: the Biology and Ecology of Non-Biting Midges (Chapman & Hall, London, 1995).
Echols, H., Garen, A., Garen, S. & Torriani, A. J. Mol. Biol. 3, 425–438 (1961).
Lewin, S. M. in Cholera and the Ecology of Vibrio cholerae (eds Drasar, B. S. & Forrest, B. D.) 228–254 (Chapman & Hall, London, 1996).
Colwell, R. R. & Huq, A. in Vibrio cholerae and Cholera: Molecular to Global Perspective (eds Wachsmuth, I. K., Blake, P. A. & Olsvik, Ø.) 117–133 (ASM, Washington DC, 1994).
Roszak, D. B. & Colwell, R. R. Microbiol. Rev. 51, 365–379 (1987).
Colwell, R. R. Science 274, 2025–2031 (1996).
Colwell, R. R. & Huq, A. J. Appl. Microsc. Symp. 85 (suppl.), 134–137 (1999).
Harvell, C. D. et al. Science 285, 1505–1510 (1999).
Nolte, U. Ent. Scand. Suppl. 43, 5–75 (1993).
Broza, M., Halpern, M. & Inbar, M. Wat. Sci.Technol. 42, 71–74 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Broza, M., Halpern, M. Chironomid egg masses and Vibrio cholerae. Nature 412, 40 (2001). https://doi.org/10.1038/35083691
Issue Date:
DOI: https://doi.org/10.1038/35083691
This article is cited by
-
Vibrio cholerae O1 Inhabit Intestines and Spleens of Fish in Aquaculture Ponds
Microbial Ecology (2024)
-
Hemocytes of a tropical midge Chironomus ramosus (Diptera: Chironomidae)
International Journal of Tropical Insect Science (2024)
-
Mining the diversity and functional profile of bacterial symbionts from the larvae of Chironomus circumdatus (bloodworms)
Folia Microbiologica (2022)
-
Beyond the swab: ecosystem sampling to understand the persistence of an amphibian pathogen
Oecologia (2018)
-
Great cormorants (Phalacrocorax carbo) as potential vectors for the dispersal of Vibrio cholerae
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.