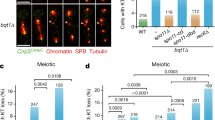
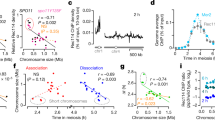
Abstract
Meiosis is initiated from G1 of the cell cycle and is characterized by a pre-meiotic S phase followed by two successive nuclear divisions. The first of these, meiosis I, differs from mitosis in having a reductional pattern of chromosome segregation1,2. Here we show that meiosis can be initiated from G2 in fission yeast cells by ectopically activating the meiosis-inducing network. The subsequent meiosis I occurs without a pre-meiotic S phase and with decreased recombination, and exhibits a mitotic pattern of equational chromosome segregation. The subsequent meiosis II results in random chromosome segregation. This behaviour is similar to that observed in cells lacking the meiotic cohesin Rec8 (refs 3, 4), which becomes associated with chromosomes at G1/S phase, including the inner centromere, a region that is probably critical for sister-centromere orientation5. If the expression of Rec8 is delayed to S phase/G2, then the centromeres behave equationally. We propose that the presence of Rec8 in chromatin is required at the pre-meiotic S phase to construct centromeres that behave reductionally and chromosome arms capable of a high level of recombination, and that this explains why meiosis is initiated from G1 of the cell cycle.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kleckner, N. Meiosis: how could it work? Proc. Natl Acad. Sci. USA 93, 8167–8174 (1996).
Roeder, G. S. Meiotic chromosomes: it takes two to tango. Genes Dev. 11, 2600–2621 (1997).
DeVeaux, L. C. & Smith, G. R. Region-specific activators of meiotic recombination in Schizosaccharomyces pombe. Genes Dev. 8, 203–210 (1994).
Watanabe, Y. & Nurse, P. Cohesin Rec8 is required for reductional chromosome segregation at meiosis. Nature 400, 461–464 (1999).
Takahashi, K., Chen, E. S. & Yanagida, M. Requirement of Mis6 centromere connector for localizing a CENP-A-like protein in fission yeast. Science 288 , 2215–2219 (2000).
Iino, Y. & Yamamoto, M. Negative control for the initiation of meiosis in Schizosaccharomyces pombe. Proc. Natl Acad. Sci. USA 82, 2447–2451 ( 1985).
McLeod, M. & Beach, D. A specific inhibitor of the ran1 + protein kinase regulates entry into meiosis in Schizosaccharomyces pombe. Nature 332, 509– 514 (1988).
Watanabe, Y., Shinozaki-Yabana, S., Chikashige, Y., Hiraoka, Y. & Yamamoto, M. Phosphorylation of RNA-binding protein controls cell cycle switch from mitotic to meiotic in fission yeast. Nature 386, 187–190 (1997).
Yamamoto, M., Imai, Y. & Watanabe, Y. in The Molecular and Cellular Biology of the Yeast Saccharomyces (eds Pringle, J., Broach, J. & Jones, E.) 1037– 1106 (Cold Spring Harbor Laboratory, Cold Spring Harbor, New York, 1997).
Horie, S. et al. The Schizosaccharomyces pombe mei4+ gene encodes a meiosis-specific transcription factor containing a forkhead DNA-binding domain. Mol. Cell. Biol. 18, 2118– 2129 (1998).
Bähler, J., Schuchert, P., Grimm, C. & Kohli, J. Synchronized meiosis and recombination in fission yeast: observations with pat1-114 diploid cells. Curr. Genet. 19, 445– 451 (1991).
Niwa, O. & Yanagida, M. Universal and essential role of MPF/cdc2+. Nature 336, 430 ( 1988).
Nabeshima, K. et al. Dynamics of centromeres during metaphase–anaphase transition in fission yeast: Dis1 is implicated in force balance in metaphase bipolar spindle. Mol. Biol. Cell 9, 3211– 3225 (1998).
Klein, F. et al. A central role for cohesins in sister chromatid cohesion, formation of axial elements, and recombination during yeast meiosis. Cell 98, 91–103 ( 1999).
Takahashi, K. et al. A low copy number central sequence with strict symmetry and unusual chromatin structure in fission yeast centromere. Mol. Biol. Cell 3, 819–835 ( 1992).
Partridge, J. F., Borgstrom, B. & Allshire, R. C. Distinct protein interaction domains and protein spreading in a complex centromere. Genes Dev. 14, 783–791 (2000).
Toth, A. et al. Yeast cohesin complex requires a conserved protein, Eco1p(Ctf7), to establish cohesion between sister chromatids during DNA replication. Genes Dev. 13, 320–333 ( 1999).
Uhlmann, F. & Nasmyth, K. Cohesion between sister chromatids must be established during DNA replication. Curr. Biol. 8, 1095–1101 (1998).
Cha, R. S., Weiner, B. M., Keeney, S., Dekker, J. & Kleckner, N. Progression of meiotic DNA replication is modulated by interchromosomal interaction proteins, negatively by Spo11p and positively by Rec8p. Genes Dev. 14, 493–503 (2000).
Molnar, M., Bähler, J., Sipiczki, M. & Kohli, J. The rec8 gene of Schizosaccharomyces pombe is involved in linear element formation, chromosome pairing and sister-chromatid cohesion during meiosis. Genetics 141, 61– 73 (1995).
Moreno, S., Klar, A. & Nurse, P. Molecular genetic analysis of fission yeast Schizosaccharomyces pombe. Methods Enzymol. 194, 795– 823 (1991).
Saitoh, S., Takahashi, K. & Yanagida, M. Mis6, a fission yeast inner centromere protein, acts during G1/S and forms specialized chromatin required for equal segregation. Cell 90, 131–143 (1997).
Kelly, T. J. et al. The fission yeast cdc18 gene product couples S-phase to start and mitosis. Cell 74, 371– 382 (1993).
Fernandez-Sarabia, M. J., McInery, C., Harris, P., Gordon, C. & Fantes, P. The cell cycle genes cdc22+ and suc22+ of the fission yeast Schizosaccharomyces pombe encode the large and small subunits of ribonucleotide reductase. Mol. Gen. Genet. 238, 241–251 (1993).
Lin, L. & Smith, G. R. Transient, meiosis-induced expression of the rec6 and rec12 genes of Schizosaccharomyces pombe . Genetics 136, 769– 779 (1994).
Acknowledgements
We thank J. P. Cooper for critical reading of the manuscript and M. Yanagida for CHIP method. Y.W. thanks all the members of P.N.'s laboratory for help and discussion, particularly J. Hayles, H. Murakami and G. Simchen. Y.W. was supported by JSPS and Uehara fellowships and grants from the Ministry of Education, Science and Culture of Japan.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Watanabe, Y., Yokobayashi, S., Yamamoto, M. et al. Pre-meiotic S phase is linked to reductional chromosome segregation and recombination. Nature 409, 359–363 (2001). https://doi.org/10.1038/35053103
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35053103
This article is cited by
-
Transient reduction of DNA methylation at the onset of meiosis in male mice
Epigenetics & Chromatin (2018)
-
Chromosome pairing in meiosis of partially fertile wheat/rye hybrids
Plant Reproduction (2013)
-
Geometry and force behind kinetochore orientation: lessons from meiosis
Nature Reviews Molecular Cell Biology (2012)
-
OsAM1 is required for leptotene-zygotene transition in rice
Cell Research (2011)
-
Acetylation regulates monopolar attachment at multiple levels during meiosis I in fission yeast
EMBO reports (2011)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.