Abstract
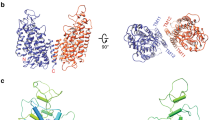
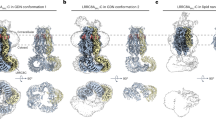
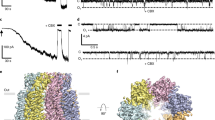
Virtually all cells in all eukaryotic organisms express ion channels of the ClC type, the only known molecular family of chloride-ion-selective channels. The diversity of ClC channels highlights the multitude and range of functions served by gated chloride-ion conduction in biological membranes, such as controlling electrical excitability in skeletal muscle, maintaining systemic blood pressure, acidifying endosomal compartments, and regulating electrical responses of GABA (γ-aminobutyric acid)-containing interneurons in the central nervous system1. Previously, we expressed and purified a prokaryotic ClC channel homologue2. Here we report the formation of two-dimensional crystals of this ClC channel protein reconstituted into phospholipid bilayer membranes. Cryo-electron microscopic analysis of these crystals yields a projection structure at 6.5 Å resolution, which shows off-axis water-filled pores within the dimeric channel complex.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Maduke, M., Miller, C. & Mindell, J. A. A decade of CLC chloride channels: structure, mechanism, and many unsettled questions. Annu. Rev. Biophys. Biomol. Struct. 29, 411–438 ( 2000).
Maduke, M., Pheasant, D. J. & Miller, C. High-level expression, functional reconstitution, and quaternary structure of a prokaryotic ClC-type chloride channel. J. Gen. Physiol. 114, 713–722 (1999).
Jentsch, T. J. & Gunther, W. Chloride channels: an emerging molecular picture. BioEssays 19, 117– 126 (1997).
Ludewig, U., Pusch, M. & Jentsch, T. J. Two physically distinct pores in the dimeric ClC-0 chloride channel. Nature 383, 340– 343 (1996).
Ludewig, U., Pusch, M. & Jentsch, T. J. Independent gating of single pores in CLC-0 chloride channels. Biophys. J. 73, 789– 797 (1997).
Miller, C. Open-state substructure of single chloride channels from Torpedo electroplax. Phil. Trans. R. Soc. Lond. B 299, 401– 411 (1982).
Middleton, R. E., Pheasant, D. J. & Miller, C. Homodimeric architecture of a ClC-type chloride ion channel. Nature 383, 337– 340 (1996).
Saviane, C., Conti, F. & Pusch, M. The muscle chloride channel ClC-1 has a double-barreled appearance that is differentially affected in dominant and recessive myotonia. J. Gen. Physiol. 113, 457– 468 (1999).
Fahlke, C., Rhodes, T. H., Desai, R. R. & George, A. L. Jr. Pore stoichiometry of a voltage-gated chloride channel. Nature 394, 687–690 ( 1998).
Valpuesta, J. M., Carrascosa, J. L. & Henderson, R. Analysis of electron microscope images and electron diffraction patterns of thin crystals of phi 29 connectors in ice. J. Mol. Biol. 240, 281–287 (1994).
Middleton, R. E., Pheasant, D. J. & Miller, C. Purification, reconstitution, and subunit composition of a voltage-gated chloride channel from Torpedo electroplax. Biochemistry 33, 13189–13198 (1994).
Grigorieff, N., Beckmann, E. & Zemlin, F. Lipid location in deoxycholate-treated purple membrane at 2.6 Å. J. Mol. Biol. 254, 404–415 (1995).
Jentsch, T. J., Steinmeyer, K. & Schwarz, G. Primary structure of Torpedo marmorata chloride channel isolated by expression cloning in Xenopus oocytes. Nature 348, 510–514 ( 1990).
Henderson, R., Baldwin, J. M., Downing, K. H., Lepault, J. & Zemlin, F. Structure of purple membrane from halobacterium halobium: recording, measurement and evaluation of electron micrographs at 3.5 Å resolution. Ultramicroscopy 19, 147–178 (1986).
Crowther, R. A., Henderson, R. & Smith, J. M. MRC image processing programs. J. Struct. Biol. 116, 9–16 ( 1996).
Schertler, G. F., Villa, C. & Henderson, R. Projection structure of rhodopsin. Nature 362, 770–772 ( 1993).
Acknowledgements
We are grateful to R. Henderson for making facilities at the MRC, including the Hitachi microscope, available to us for low-dose experiments, to J. Berriman for help and hand-holding, and to T. Walz for crucial advice on crystallization. We also acknowledge D. Pheasant and L. Melanson for technical assistance. Support was provided in part by a grant to J.A.M. from the NIH.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mindell, J., Maduke, M., Miller, C. et al. Projection structure of a ClC-type chloride channel at 6.5 Å resolution. Nature 409, 219–223 (2001). https://doi.org/10.1038/35051631
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35051631
This article is cited by
-
Unfolding of a ClC chloride transporter retains memory of its evolutionary history
Nature Chemical Biology (2018)
-
ClC chloride channels viewed through a transporter lens
Nature (2006)
-
Ion channels and ion transporters of the transverse tubular system of skeletal muscle
Journal of Muscle Research and Cell Motility (2006)
-
Electron and atomic force microscopy of the trimeric ammonium transporter AmtB
EMBO reports (2004)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.