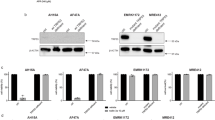
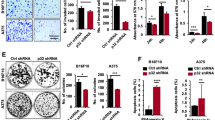
Abstract
Metastatic melanoma is a deadly cancer that fails to respond to conventional chemotherapy and is poorly understood at the molecular level1. p53 mutations often occur in aggressive and chemoresistant cancers but are rarely observed in melanoma1,2. Here we show that metastatic melanomas often lose Apaf-1, a cell-death effector that acts with cytochrome c and caspase-9 to mediate p53-dependent apoptosis3. Loss of Apaf-1 expression is accompanied by allelic loss in metastatic melanomas, but can be recovered in melanoma cell lines by treatment with the methylation inhibitor 5-aza-2′-deoxycytidine (5aza2dC). Apaf-1-negative melanomas are invariably chemoresistant and are unable to execute a typical apoptotic programme in response to p53 activation. Restoring physiological levels of Apaf-1 through gene transfer or 5aza2dC treatment markedly enhances chemosensitivity and rescues the apoptotic defects associated with Apaf-1 loss. We conclude that Apaf-1 is inactivated in metastatic melanomas, which leads to defects in the execution of apoptotic cell death. Apaf-1 loss may contribute to the low frequency of p53 mutations observed in this highly chemoresistant tumour type.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Chin, L., Merlino, G. & DePinho, R. A. Malignant melanoma: modern black plague and genetic black box. Genes Dev. 12, 3467– 3481 (1998).
Serrone, L. & Hersey, P. The chemoresistance of human malignant melanoma: an update. Melanoma Res. 9, 51 –58 (1999).
Soengas, M. S. et al. Apaf-1 and caspase-9 in p53-dependent apoptosis and tumor inhibition. Science 284, 156– 159 (1999).
Schmitt, C. A. & Lowe, S. W. Apoptosis and therapy. J. Pathol. 187, 127–137 (1999).
Lowe, S. W., Ruley, H. E., Jacks, T. & Housman, D. E. p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74, 957–967 ( 1993).
Hakem, R. et al. Differential requirement for caspase 9 in apoptotic pathways in vivo. Cell 94, 339– 352 (1998).
Fearnhead, H. O. et al. Oncogene-dependent apoptosis is mediated by caspase-9. Proc. Natl Acad. Sci. USA 95, 13664– 13669 (1998).
Baylin, S. B. & Herman, J. G. DNA hypermethylation in tumorigenesis: epigenetics joins genetics. Trends Genet. 16, 168–174 (2000).
Robertson, K. D. & Jones, P. A. DNA methylation: past, present and future directions. Carcinogenesis 21, 461–467 (2000).
Bird, A. P. & Wolffe, A. P. Methylation-induced repression—belts, braces, and chromatin. Cell 99, 451– 454 (1999).
Hark, A. T. et al. CTCF mediates methylation-sensitive enhancer-blocking activity at the H19/Igf2 locus. Nature 405, 486– 489 (2000).
Bell, A. C. & Felsenfeld, G. Methylation of a CTCF-dependent boundary controls imprinted expression of the Igf2 gene. Nature 405, 482–485 ( 2000).
Lowe, S. W. & Ruley, H. E. Stabilization of the p53 tumor suppressor is induced by adenovirus 5 E1A and accompanies apoptosis. Genes Dev. 7, 535–545 ( 1993).
Green, D. R. & Reed, J. C. Mitochondria and apoptosis. Science 281, 1309–1312 ( 1998).
Rodriguez, J. & Lazebnik, Y. Caspase-9 and APAF-1 form an active holoenzyme. Genes Dev. 13, 3179– 3184 (1999).
Zou, H., Li, Y., Liu, X. & Wang, X. An APAF-1 cytochrome c multimeric complex is a functional apoptosome that activates procaspase-9. J. Biol. Chem. 274, 11549– 11556 (1999).
Hu, Y., Benedict, M. A., Ding, L. & Nunez, G. Role of cytochrome c and dATP/ATP hydrolysis in Apaf-1-mediated caspase- 9 activation and apoptosis. EMBO J. 18, 3586 –3595 (1999).
Teitz, T. et al. Caspase 8 is deleted or silenced preferentially in childhood neuroblastomas with amplification of MYCN. Nature Med. 6, 529–535 (2000).
Yamamoto, H., Gil, J., Schwartz, S. Jr & Perucho, M. Frameshift mutations in Fas, Apaf-1, and Bcl-10 in gastro-intestinal cancer of the microsatellite mutator phenotype. Cell Death Differ. 7, 238–239 (2000).
Schmitt, C. A., McCurrach, M. E., de Stanchina, E., Wallace-Brodeur, R. R. & Lowe, S. W. INK4a/ARF mutations accelerate lymphomagenesis and promote chemoresistance by disabling p53. Genes Dev. 13, 2670–2677 (1999).
Chin, L. et al. Cooperative effects of INK4a and ras in melanoma susceptibility in vivo. Genes Dev. 11, 2822– 2834 (1997).
Walker, G. J. et al. Virtually 100% of melanoma cell lines harbor alterations at the DNA level within CDKN2A, CDKN2B, or one of their downstream targets. Genes Chromosom. Cancer 22, 157–163 (1998).
Grossman, D., McNiff, J. M., Li, F. & Altieri, D. C. Expression and targeting of the apoptosis inhibitor, survivin, in human melanoma. J. Invest. Dermatol. 113, 1076–1081 (1999).
Irmler, M. et al. Inhibition of death receptor signals by cellular FLIP. Nature 388, 190–195 ( 1997).
Selzer, E. et al. Expression of Bcl-2 family members in human melanocytes, in melanoma metastases and in melanoma cell lines. Melanoma Res. 8, 197–203 (1998).
Mora, J. et al. Clinical categories of neuroblastoma are associated with different patterns of loss of heterozygosity on chromosome Arm 1p. J. Mol. Diag. 2, 1–9 ( 2000).
Woo, M. et al. Essential contribution of caspase 3/CPP32 to apoptosis and its associated nuclear changes. Genes Dev. 12, 806– 819 (1998).
Marcellus, R. C., Teodoro, J. G., Charbonneau, R., Shore, G. C. & Branton, P. E. Expression of p53 in Saos-2 osteosarcoma cells induces apoptosis which can be inhibited by Bcl-2 or the adenovirus E1B-55 kDa protein. Cell Growth Differ. 7, 1643–1650 (1996).
Capodieci, P., Magi-Galluzzi, C., Moreira, G. Jr Zeheb, R. & Loda, M. Automated in situ hybridization: diagnostic and research applications. Diagn. Mol. Pathol. 7, 69–75 ( 1998).
Acknowledgements
We thank B. Stillman and N. K. Cheung for support; M. McCurrach, W. Jiang, L. Chen and M. Dudas for technical assistance; M. Zhang, R. Davuluri and I. Ioshikhes for analysis of Apaf-1 CpG islands and assembly of Apaf-1 genomic sequence; G. Núñez for Apaf-1XL pcDNA construct; E. Querido, P. E. Branton and F. L. Graham for Ad-p53 and Ad-LacZ; and A. Houghton and R. Camalier for melanoma cell lines. We also thank G. Hannon, C. Schmitt, C. Hazan, K. Pohar, T. Tiongson and the Cold Spring Harbor Genome Center for help and advice. Y.L. is a Pew Scholar and S.W.L. is a Rita Allen Scholar. This work was supported by a long-term Postdoctoral Fellowship from the Human Frontiers in Science Program and a Special Fellowship from the Leukemia and Lymphoma Society (M.S.S), a K08 grant (D.P.), an ASCO Young Investigator Award (J.M.), grants from the Seraph Foundation (YL), and from the NIH and the NCI (C.C.C. and S.W.L.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Soengas, M., Capodieci, P., Polsky, D. et al. Inactivation of the apoptosis effector Apaf-1 in malignant melanoma . Nature 409, 207–211 (2001). https://doi.org/10.1038/35051606
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35051606
This article is cited by
-
Novel meriolin derivatives activate the mitochondrial apoptosis pathway in the presence of antiapoptotic Bcl-2
Cell Death Discovery (2024)
-
Common DNA methylation dynamics in endometriod adenocarcinoma and glioblastoma suggest universal epigenomic alterations in tumorigenesis
Communications Biology (2021)
-
Low expression of pro-apoptotic proteins Bax, Bak and Smac indicates prolonged progression-free survival in chemotherapy-treated metastatic melanoma
Cell Death & Disease (2020)
-
Aldo-keto reductases protect metastatic melanoma from ER stress-independent ferroptosis
Cell Death & Disease (2019)
-
Molecular changes in solitary fibrous tumor progression
Journal of Molecular Medicine (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.