Abstract
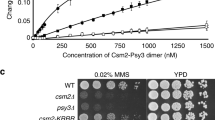
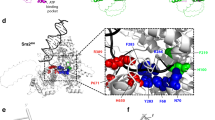
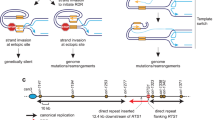
In Saccharomyces cerevisiae, the RAD51 and RAD52 genes are involved in recombination and in repair of damaged DNA1,2,3. The RAD51 gene is a structural and functional homologue of the recA gene4,5 and the gene product participates in strand exchange and single-stranded-DNA-dependent ATP hydrolysis by means of nucleoprotein filament formation6,7,8,9,10,11. The RAD52 gene12 is important in RAD51-mediated recombination1,2,3. Binding of this protein to Rad51 (refs 4, 13) suggests that they cooperate in recombination. Homologues of both Rad51 and Rad52 are conserved from yeast to humans14,15,16, suggesting that the mechanisms used for pairing homologous DNA molecules during recombination may be universal in eukaryotes. Here we show that Rad52 protein stimulates Rad51 reactions and that binding to Rad51 is necessary for this stimulatory effect. We conclude that this binding is crucial in recombination and that it facilitates the formation of Rad51 nucleoprotein filaments.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Resnick, M. A. in Meiosis (ed. Moens, P. B.) 157–210 (Academic, New York, 1987).
Petes, T. D., Malone, R. E. & Symington, L. S. in The Molecular and Cellular Biology of the Yeast Saccharomyces. Genome Dynamics, Protein Synthesis and Energetics. (eds Broach, J. R., Pringle, J. R. & Jones, E. W.) 407–521 (Cold Spring Harbor Laboratory Press, New York, 1991).
Shinohara, A. & Ogawa, T. Homologous recombination and the roles of double-strand breaks. Trends Biochem. Sci. 20, 587–591 (1995).
Shinohara, A., Ogawa, H. & Ogawa, T. Rad51 protein involved in repair and recombination in S. cerevisiae is a RecA-like protein. Cell 69, 457–470 (1992).
Aboussekhra, A., Chanet, R., Adjiri, A. & Fabre, F. Semidominant suppressors of Srs2 helicase mutations of Saccharomyces cerevisiae map in the RAD51 gene, whose sequence predicts a protein with similarities to prokaryotic RecA proteins. Mol. Cell. Biol. 12, 3224–3234 (1992).
Ogawa, T., Yu, X., Shinohara, A. & Egelman, E. H. Similarity of the yeast RAD51 filament ot the bacterial Rec filament. Science 259, 1896–1899 (1993).
Ogawa, T.et al. RecA-like recombination proteins in eukaryotes: Functions and structures of RAD51 gene. Cold Spring Harb. Symp. Quant. Biol. 58, 567–576 (1994).
Sung, P. Catalysis of ATP-dependent homologous DNA pairing and strand exchange by yeast RAD51 protein. Science 265, 1241–1243 (1994).
Sung, P. & Robberson, D. L. DNA strand exchange mediated by a RAD51–ssDNA nucleoprotein filament with polarity opposite to that of RecA. Cell 82, 453–461 (1995).
Sugiyama, T., Zaitseva, E. M. & Kowalczykowski, S. C. Asingle-stranded DNA binding protein is needed for efficient presynaptic complex formation by the Saccharomyces cerevisiae Rad51 proteins. J. Biol. Chem. 272, 7940–7945 (1997).
Namsaraev, E. & Berg, P. Characterization of strand exchange activity of yeast Rad51 protein. Mol. Cell. Biol. 17, 5359–5368 (1997).
Adzuma, K., Ogawa, T. & Ogawa, H. Primary structure of the RAD52 gene in Saccharomyces cerevisiae. Mol. Cell. Biol. 4, 2735–2744 (1984).
Milne, G. T. & Weaver, D. T. Dominant negative alleles of RAD52 reveal a DNA repair/recombination complex including Rad51 and Rad52. Genes Dev. 7, 1755–1765 (1993).
Shinohara, A.et al. Cloning of human, mouse and fission yeast recombination genes homologous to RAD51 and recA. Nature Genet. 4, 239–243 (1993).
Murris, D. F. R.et al. Cloning of human and mouse genes homologous to RAD52, a yeast gene involved in DNA repair and recombination. Mutat. Res. DNA Repair 315, 295–305 (1994).
Ogawa, T., Shinohara, A. & Ikeya, T. Aspecies-specific interaction of Rad51 and Rad52 proteins in eukaryotes. Adv. Biophys. 31, 93–100 (1995).
Mortensen, U. H., Bendixen, C., Sunjevaric, I. & Rothstein, R. DNA strand annealing is promoted by the yeast Rad52 protein. Proc. Natl Acad. Sci. USA 93, 10729–10734 (1996).
Sung, P. Yeast Rad55 and Rad57 proteins form a heterodimer that functions with replication protein A to promote DNA strand exchange by Rad51 recombinase. Genes Dev. 11, 1111–1121 (1997).
Kowalczykowski, S. C. & Eggleston, A. K. Homologous pairing and DNA strand exchange proteins. Annu. Rev. Biochem. 63, 991–1043 (1994).
West, S. C. Enzymes and molecular mechanisms of genetic recombination. Annu. Rev. Biochem. 61, 603–640 (1992).
Boundy-Mills, K. L. & Livingston, D. M. A Saccharomyces cerevisiae RAD52 allele expressing a C-terminal truncation protein: Activities and intragenic complementation of missense mutations. Genetics 133, 39–49 (1993).
Baumann, P., Benson, F. E. & West, S. C. Human Rad51 protein promotes ATP-dependent homologous pairing and strand transfer reactions in vitro. Cell 87, 757–766 (1996).
Gupta, R. C., Bazemore, L. R., Golub, E. I. & Radding, C. M. Activities of human recombination protein Rad51. Proc. Natl Acad. Sci. USA 94, 463–468 (1997).
Shen, Z., Cloud, K. G., Chen, D. J. & Park, M. S. Specific interactions between the human RAD51 and RAD52 proteins. J. Biol. Chem. 271, 148–152 (1996).
Shinohara, A., Shinohara, M., Ohta, T., Matsuda, S. & Ogawa, T. Cooperation of Rad52 with a single-stranded DNA binding protein, RPA, in homologous recombination. Genes-to-Cells(submitted).
Alani, E., Thresher, R., Griffith, J. D. & Kolodner, R. D. Characterization of DNA-binding and strand-exchange stimulation properties of y-RPA, a yeast single-strand-DNA-binding protein. J. Mol. Biol. 227, 54–71 (1992).
Benson, F. E., Baumann, P. & West, S. C. Synergistic actions of Rad51 and Rad52 in recombination and DNA repair. Nature 391, 401–404 (1998).
New, J. H., Sugiyama, T., Zaitseva, E. & Kowalczykowski, S. C. Rad52 protein stimulates DNA strand exchange by Rad51 protein and replication protein A. Nature 391, 407–410 (1998).
Sung, P. Functions of yeast Rad52 protein as a mediator between Replication protein A and the rad51 recombinase. J. Biol. Chem. 272, 28194–28197 (1997).
Acknowledgements
We thank J. Tomizawa and H. Ogawa for encouragement and support; N. Kleckner, D. Bishop, E. Egelman, L. S. Symington and S. Gaisor for critically reading the manuscript; A. Sugino for yeast RPA protein; R. Kolodner for the overproducer strain of RPA; and D. Bishop and S. West for communicating unpublished results. This work was supported by grants from the Ministry of Education, Science, Sports and Culture of Japan and the Human Frontier Science Organization to A.S. and T.O., and a grant from National Institute of Genetics to A.S.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Shinohara, A., Ogawa, T. Stimulation by Rad52 of yeast Rad51- mediated recombination. Nature 391, 404–407 (1998). https://doi.org/10.1038/34943
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/34943
This article is cited by
-
FIGNL1 AAA+ ATPase remodels RAD51 and DMC1 filaments in pre-meiotic DNA replication and meiotic recombination
Nature Communications (2023)
-
Bre1/RNF20 promotes Rad51-mediated strand exchange and antagonizes the Srs2/FBH1 helicases
Nature Communications (2023)
-
Expression of human BRCA2 in Saccharomyces cerevisiae complements the loss of RAD52 in double-strand break repair
Current Genetics (2023)
-
Structural basis of homologous recombination
Cellular and Molecular Life Sciences (2020)
-
Dynamics and selective remodeling of the DNA-binding domains of RPA
Nature Structural & Molecular Biology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.