Abstract
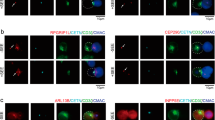
CELLULARras genes encode a family of membrane-associated proteins (p21ras) that bind guanine nucleotide and possess a low intrinsic GTPase activity1–3. The p21ras proteins are ubiquitously expressed in mammalian cells and are thought to be involved in a growth-promoting signal transduction pathway4; their mode of action, however, remains unknown. The ligand-induced movement of cell-surface receptors seems to be a primary event in the transduction of several extracellular signals that control cell growth and differentiation. In B lymphocytes, surface immunoglobulin receptors crosslinked by antibody or other multivalent ligands form aggregates called patches, which then collect into a single assembly, a cap, at one pole of the cell5,6. This process constitutes the initial signal for the activation of a B cell. Here we show by immunofluorescence microscopy that p21ras co-caps with surface immunoglobulin molecules in mouse splenic B lymphocytes. In contrast, no apparent change in the distribution of p21ras occurs during the capping of concanavalin A receptors. The redistribution of p21ras is apparent at the early stages (patching) of immunoglobulin capping and is inhibited by metabolic inhibitors and the cytoskeleton-disrupting agents colchicine and cytochalasin D. The distribution of another membrane-associated guanine nucleotide-binding regulatory protein, the Giα subunit, is not affected by surface immunoglobulin capping. These findings demonstrate that p2lras can migrate in a directed manner along the plasma membrane and suggest that p21ras may be a component of the signalling pathway initiated by the capping of surface immunoglobulin in B lymphocytes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
McGrath, J. P., Capon, D. J., Goeddel, D. V. & Levinson, A. D. Nature 310, 644–649 (1984).
Sweet, R. W. et al. Nature 311, 273–275 (1984).
Gibbs, J. B., Sigal, I. S., Poe, M. & Scolnick, E. M. Proc. natn. Acad. Sci. U.S.A. 81, 5704–5708 (1984).
Barbacid, M. A. Rev. Biochem. 56, 779–827 (1987).
Taylor, R. B., Duffus, P. H., Raff, M. C. & dePetris, S. Nature new Biol. 233, 225–229 (1971).
Unanue, E. R., Perkins, W. D. & Karnovsky, M. J. J. exp. Med. 136, 885–897 (1972).
Gross, M. et al. Molec. cell. Biol. 5, 1015–1024 (1985).
Willingham, M. C., Pastan, I., Shin, T. C. & Scolnick, E. M. Cell 19, 1005–1014 (1980).
Scribner, D. J., Weiner, H. L. & Moorhead, J. W. J. Immun. 121, 377–383 (1978).
Philips, N. E. & Parker, D. C. J. Immun. 132, 627–632 (1984).
Bokoch, G. M., Bickford, K. & Bohl, B. P. J. Cell Biol. 106, 1927–1936 (1988).
Unanue, E. R., Karnovsky, M. J. & Engers, H. D. J. exp. Med. 137, 675–689 (1973).
Loor, F., Forni, L. & Pernis, B. Eur. J. Immun. 2, 203–212 (1972).
Schreiner, G. F., Fujiwara, K., Pollard, T. D. & Unanue, E. R. J. exp. Med. 145, 1393–1398 (1977).
Gabbiani, G., Chaponnier, C., Zumbe, A. & Bassali, P. Nature 269, 697–698 (1977).
Bourguignon, L. Y. W., Tokuyasu, K. T. & Singer, S. J. J. cell Physiol. 95, 239–258 (1978).
Edelman, G. M., Yahara, I. & Wang, J. L. Proc. natn. Acad. Sci. U.S.A. 70, 1442–1446 (1973).
dePetris, S. Nature 250, 54–56 (1974).
Wessels, N. K. et al. Science 171, 135–143 (1971).
Borisy, G. G. & Taylor, E. W. J. cell Biol. 34, 525–534 (1967).
Geiger, B. & Singer, S. J. Cell 16, 213–222 (1979).
Weihung, R. R. in Methods and Achievements in Experimental Biology Vol. 8 (ed. Gabbiani, G.) 42–109 (Kargard, New York, 1979).
Gold, M. R., Jakway, J. P. & Defranco, A. L. J. Immun. 139, 3604–3613 (1987).
Harnett, M. M. & Klaus, G. G. B. J. Immun. 140, 3135–3139 (1988).
Monroe, J. & Haldar, S. Biochim. biophys. Acta 1013, 273–278 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Graziadei, L., Riabowol, K. & Bar-Sagi, D. Co-capping of ras proteins with surface immunoglobulins in B lymphocytes. Nature 347, 396–400 (1990). https://doi.org/10.1038/347396a0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1038/347396a0
This article is cited by
-
Monte Carlo study of B-cell receptor clustering mediated by antigen crosslinking and directed transport
Cellular & Molecular Immunology (2011)
-
TRAIL activates acid sphingomyelinase via a redox mechanism and releases ceramide to trigger apoptosis
Oncogene (2006)
-
Initiation of antigen receptor endocytosis and B lymphocyte activation lie on independent biochemical pathways
Immunology & Cell Biology (1993)
-
Antigen Responsive Antibody–Receptor Kinase Chimera
Bio/Technology (1992)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.