Abstract
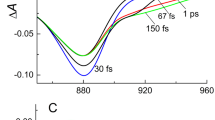
The conversion of light energy to chemical energy during photosyn-thesis involves the transfer of electrons between pigments embedded in a membrane protein. This process occurs with high quantum efficiency, the result of extremely fast electron transfer over a long distance preventing back transfer and energy loss. Recently the three-dimensional structures of the photosynthetic reaction centres of the bacteria Rhodopseudomonas viridis1 and Rhodobacter sphaeroides2 have been determined, allowing a molecular descrip-tion of the primary charge separation process. There are two symmetrically related branches of pigments in the structure (L and M), extending from the special pair of bacteriochlorophyll molecules (P) to the two bacteriopheophytins (HL and HM) via two bacteriochlorophylls (BLand BM). Many features of the electron transfer process are poorly understood, such as the nature of the excited states involved, the identity of the primary charge separation step and the roles of the protein and of B3–13. We have determined the rates of electron transfer in isolated reaction centre complexes of Rps. viridis and Rb. sphaeroides as a function of temperature. The rates increase as temperature is decreased, which may be due to either changes in electronic coupling of the pigments or changes in the population of coupled vibrational modes, or a combination of the two. We see no evidence of a B−L intermediate, which sets a lower limit on the rate of electron transfer from BL to HL. This is so high as to rule out transfer by two non-adiabatic steps.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Deisenhofer, J., Epp, O., Miki, K., Huber, R. & Michel, H. J. molec. Biol. 180, 385–398 (1984).
Allen, J. P., Feher, G., Yeates, T. O., Komiya, H. & Rees, D. C. Proc. natn. Acad. Sci. U.S.A. 84, 5730–5734 (1987).
Scherer, P. O. J. & Fisher, S. F. Biochim. biophys. Acta 891, 157–164 (1987).
Parson, W. W. & Warshel, A. J. Am. chem. Soc. 109, 6152–6163 (1987).
Lous, E. J. & Hoff, A. J. Proc. natn. Acad. Sci. U.S.A. 84, 6147–6151 (1987).
Won, Y. & Friesner, R. A. Proc. natn. Acad. Sci. U.S.A. 84, 5511–5514 (1987).
Marcus, R. A. Chem. Phys. Lett. 133, 471–477 (1987).
Parson, W. W., Scherz, A. & Warshel, A. in Antennas and Reaction Centers of Photosynthetic Bacteria (ed. M E. Michel-Beyerle) 122–126 (Springer, Berlin, 1985).
Bixon, M., Jortner, J., Michel-Beyerle, M. E., Ogrodnik, A. & Lersch, W. Chem. Phys. Lett. 140, 626–630 (1987).
Chekalin, S. V., Matveetz, Ya. A., Shkuropatov, A. Ya., Shuvalov, V. A. & Yartzev, A. P. FEBS Lett. 216, 245–248 (1987).
Bixon, M. & Jortner, J. J. phys. Chem. 90, 3795–3800 (1986).
Marcus, R. A. & Sutin, N. Biochim. biophys. Acta 811, 265–322 (1985).
Friesner, R. & Wertheimer, R. Proc. natn. Acad. Sci. U.S.A. 79, 2138–2142 (1982).
Martin, J. L., Breton, J., Hofl, A. J., Migus, A. & Antonetti, A. Proc. natn. Acad. Sci. U.S.A. 83, 957–961 (1986).
Breton, J., Martin, J. L., Migus, A., Antonetti, A. & Orszag, A. Proc. natn. Acad. Sci. U.S.A. 83, 5121–5125 (1986).
Woodbury, N. W., Becker, M., Middendorf, D. & Parson, W. W. Biochemistry 24, 7516–7521 (1985).
Ditson, S. L., Davis, R. C. & Pearlstein, R. M. Biochim. biophys. Acta 766, 623–629 (1984).
Kirmaier, C., Holten, D. & Parson, W. W. Biochim. biophys. Acta 810, 33–48 (1985).
Ogrodnik, A., Remy-Richler, N. & Michel-Beyerle, M. E. Chem. Phys. Lett. 135, 576–581 (1987).
Frauenfelder, H. et al. Biochemistry 26, 254–261 (1987).
Tiede, D. M. et al. in The Photosynthetic Bacterial Reaction Center. Structure and Dynamics (eds Breton, J. & Vermeglio, A.) NATO AS1 Series (Plenum, New York, in the press).
Boxer, S. G., Lockhart, D. & Middendorf, T. R. Chem. Phys. Lett. 123, 476–482 (1986).
Meech, S. R., Hoff, A. J. & Wiersma, D. A. Chem. Phys. Lett. 121, 287–292 (1985).
Hayes, J. M. & Small, G. J. J. phys. Chem. 90, 4928–4931 (1986).
Warshel, A. Proc. natn. Acad. Sci. U.S.A. 77, 3105–3109 (1980).
Miller, J. R. in Antennas and Reaction Centers of Photosynthetic Bacteria (ed. Michel-Beyerle, M. E.) 234–241 (Springer, Berlin, 1985).
Axup, A. W., Albin, M., Mayo, S. L., Crutchley, R. J. & Gray, H. B. J. Am. chem. Soc. 140, 626–630 (1988).
Martin, J. L., Breton, J., Lambry, J. C. & Fleming, G. R. in The Photosynthetic Bacterial Reaction Center. Structure and Dynamics. (eds Breton, J. & Vermeglio, A.) NATO ASI Series (Plenum, New York, in the press).
Marcus, R. A. ibid..
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fleming, G., Martin, J. & Breton, J. Rates of primary electron transfer in photosynthetic reaction centres and their mechanistic implications. Nature 333, 190–192 (1988). https://doi.org/10.1038/333190a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/333190a0
This article is cited by
-
Ultrafast structural changes within a photosynthetic reaction centre
Nature (2021)
-
Tribute in memory of Jacques Breton (1942–2018)
Photosynthesis Research (2019)
-
Uphill energy transfer in photosystem I from Chlamydomonas reinhardtii. Time-resolved fluorescence measurements at 77 K
Photosynthesis Research (2018)
-
Effects of acid concentration on intramolecular charge transfer reaction of 4-(azetidinyl) benzonitrile in solution
Journal of Chemical Sciences (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.