Abstract
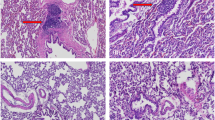
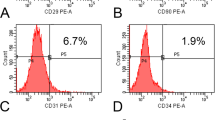
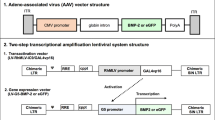
Large bone defects resulting from nonunion fractures or tumour resections are common clinical problems. Recent studies have shown bone morphogenetic protein-2 (BMP-2) gene transfer using adenoviral vectors to be a promising new therapeutic approach. However, comparative studies of different vectors are required to identify the optimal system for possible clinical trials. This study compares the use of liposome-mediated and adenoviral gene transfer for the generation of autologous BMP-2-producing bone marrow stromal cells (BMSC). Primary BMSC isolated from the rat femur were treated ex vivo with either an adenovirus or a liposome carrying human BMP-2 cDNA. The genetically modified cells were evaluated in vitro and transplanted into critical size defects in the rat mandible in vivo. BMSC treated with a reporter gene vector or untreated BMSC served as controls. The newly formed tissue was analysed by in situ hybridization, radiography and immunohistochemistry. Both groups of genetically modified cells produced BMP-2 for at least 2 weeks, and markers of new bone matrix such as osteopontin and osteocalcin were observed within 2 weeks following gene transfer. In the liposome group, the critical size defects were found completely healed at 6 weeks after the gene transfer, whereas the more efficient adenoviral gene transfer allowed for complete bone healing within 4 weeks. None of the three control groups showed bone healing, not even after 8 weeks. Thus, both liposome-mediated and adenoviral BMP-2 gene transfer to primary BMSC are suitable methods to achieve the healing of critical size bone defects in rats. As liposomes have proven sufficient for this purpose and offer several advantages over any other vector, such as ease of preparation, theoretically no limitation of the size of the DNA, and less immunological and safety problems, they may represent the best vector system for future clinical trials of bone regeneration by BMP-2 gene therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Finkemeier CG . Bone-grafting and bone-graft substitutes. J Bone Joint Surg Am 2002; 84: 454–464.
Wiltfang J et al. Degradation characteristics of alpha and beta tri-calcium-phosphate (TCP) in minipigs. J Biomed Mater Res 2002; 63: 115–121.
Murphy WL, Mooney DJ . Controlled delivery of inductive proteins, plasmid DNA and cells from tissue engineering matrices. J Periodontal Res 1999; 34: 413–419.
Perka C et al. Segmental bone repair by tissue-engineered periosteal cell transplants with bioresorbable fleece and fibrin scaffolds in rabbits. Biomaterials 2000; 21: 1145–1153.
Wozney JM et al. Novel regulators of bone formation: molecular clones and activities. Science 1988; 242: 1528–1534.
Wang EA et al. Recombinant human bone morphogenetic protein induces bone formation. Proc Natl Acad Sci 1990; 87: 2220–2224.
Cook SD, Wolfe MW, Salkeld SL, Rueger DC . Effect of recombinant human osteogenic protein-1 on healing of segmental defects in non-human primates. J Bone Joint Surg Am 1995; 77: 734–750.
Riley EH et al. Bone morphogenetic protein-2: biology and applications [Review]. Clin Orthop 1996; 324: 39–46.
Cheng SL et al. In vitro and in vivo induction of bone formation using a recombinant adenoviral vector carrying the human BMP-2 gene. Calcif Tissue Int 2001; 68: 87–94.
Mehrara B J et al. Adenovirus-mediated gene therapy of osteoblasts in vitro and in vivo. J Bone Miner Res 1999; 14: 1290–1301.
Olmsted EA et al. Adenovirus-mediated BMP-2 expression in human bone marrow stromal cells. J Cell Biochem 2001; 82: 11–21.
Riew KD et al. Induction of bone formation using a recombinant adenoviral vector carrying the human BMP-2 gene in a rabbit spinal fusion model. Calcif Tissue Int 1998; 63: 357–360.
Lieberman JR et al. Regional gene therapy with a BMP-2-producing murine stromal cell line induces heterotopic and orthotopic bone formation in rodents. J Orthop Res 1998; 16: 330–339.
Lieberman JR et al. The effect of regional gene therapy with bone morphogenetic protein-2 producing bone-marrow cells of repair of segmental femoral defects in rats. J Bone Jt Surg 1999; 81: 905–917.
Musgrave DS et al. Adenovirus-mediated direct gene therapy with bone morphogenetic protein-2 produces bone. Bone 1999; 24: 541–547.
Oakes DA, Lieberman JR . Osteoinductive applications of regional gene therapy: ex vivo gene transfer. Clin Orthop 2000; 379S:101–112.
Baltzer AW et al. Genetic enhancement of fracture repair: healing of an experimental segmental defect by adenoviral transfer of the BMP-2 gene. Gene Ther 2000; 7: 734–739.
Breitbart AS et al. Gene-enhanced tissue engineering: applications for bone healing using cultured periosteal cells transduced retrovirally with the BMP-7 gene. Ann Plast Surg 1999; 42: 488–495.
Engstrand T et al. Transient production of bone morphogenetic protein 2 by allogeneic transplanted transduced cells induces bone formation. Hum Gene Ther 2000; 11: 205–211.
Verma IM, Somia N . Gene therapy – promises, problems and prospects. Nature 1997; 389: 239–242.
Wang EA, Israel DI, Kelly S, Luxenberg DP . Bone morphogenetic protein-2 causes commitment and differentiation in C3H10T1/2 and 3T3 cells. Growth Factors 1993; 9: 57–71.
Gazit D et al. Engineered pluripotent mesenchymal cells integrate and differentiate in regenerating bone: a novel cell-mediated gene therapy. J Gene Med 1999; 1: 121–133.
Gelse K et al. Fibroblast-mediated delivery of growth factor complementary DNA into mouse joints induces chondrogenesis but avoids the disadvantages of direct viral gene transfer. Arthritis Rheum 2001; 43: 1156–1164.
Gelse K et al. Articular cartilage repair in a rat cartilage defect model by cell-mediated gene therapy using growth factor-producing mesenchymal cells. Arthritis Rheum 2003; 48: 430–441.
Kaban LB, Glowacki J . Induced osteogenesis in the repair of experimental bony defects in rats. J Dent Res 1981; 60: 1356.
Kaban LB, Glowacki J, Murray JE . Repair of experimental bony defects in rats. Surg Forum 1979; 30: 519.
Schmitz JP, Hollinger JO . The critical size defect as an experimental model for craniomandibulofacial nonunions. Clin Orthop 1986; 205: 299–308.
Lorente CA, Song BZ, Donoff RB . Healing of bony defects in the irradiated and unirradiated rat mandible. J Oral Maxillofac Surg 1992; 50: 1305–1309.
Higuchi T et al. Bone regeneration by recombinant human bone morphogenetic protein-2 in rat mandibular defects. An experimental model of defect filling. J Periodontol 1999; 70: 1026–1031.
Alden TD et al. The use of bone morphogenetic protein gene therapy in craniofacial bone repair. J Craniofac Surg 2000; 11: 24–30.
Moutsatsos IK et al. Exogenously regulated stem cell-mediated gene therapy for bone regeneration. Mol Ther 2001; 3: 449–461.
Lane JM et al. Bone marrow and recombinant human bone morphogenetic protein-2 in osseous repair. Clin Orthop 1999; 361: 216.
Sellers RS et al. Repair of articular cartilage defects one year after treatment with recombinant human bone morphogenetic protein-2 (rhBMP-2). J Bone Jt Surg 2000; 82A: 151.
Takushima A, Kitano Y, Harii K . Osteogenic potential of cultured periosteal cells in a distracted bone gap in rabbits. J Surg Res 1998; 78: 68–77.
Perka C et al. Segmental bone repair by tissue-engineered periosteal cell transplants with bioresorbable fleece and fibrin scaffolds in rabbits. Biomaterials 2000; 21: 1145–1153.
Hughes SS et al. Shared phenotypic expression of osteoblasts and chondrocytes in fracture callus. J Bone Miner Res 1995; 10: 533–544.
Nishimura T, Simmons DJ, Mainous EG . The origin of bone formed by heterotopic periosteal autografts. J Oral Maxillofac Surg 1997; 55: 1265–1268.
Harada K, Oida S, Sasaki S . BMP-2 can stimulate the bone marrow stromal cell to differentiate as osteoblast or chondroblast. Bone 1988; 9: 177–183.
Harada K et al. BMP-2 induction and TGF-beta 1 modulation of rat periosteal cell chondrogenesis. J Cell Biochem 2001; 81: 284–294.
Rowe NM et al. Rat mandibular distraction osteogenesis: Part I. Histologic and radiographic analysis. Plast Reconstr Surg 1998; 102: 2022–2032.
Ikegame M et al. Tensile stress induces bone morphogenetic protein 4 in preosteoblastic and fibroblastic cells, which later differentiate into osteoblasts leading to osteogenesis in the mouse calvariae in organ culture. J Bone Mineral Res 2001; 16: 24–32.
Acknowledgements
We thank Ms Heidemarie Heider for her expert technical assistance and Mrs Elisabeth Koppmann and Mrs Elisabeth Zimmermann for animal care. This work was supported by the Interdisciplinary Center for Clinical Research Grant D3 (to HS and KvdM) and ELAN Grant 01.04.12.1 from the University of Erlangen-Nuernberg.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Park, J., Ries, J., Gelse, K. et al. Bone regeneration in critical size defects by cell-mediated BMP-2 gene transfer: a comparison of adenoviral vectors and liposomes. Gene Ther 10, 1089–1098 (2003). https://doi.org/10.1038/sj.gt.3301960
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301960
Keywords
This article is cited by
-
Local delivery of recombinant human FGF7 enhances bone formation in rat mandible defects
Journal of Bone and Mineral Metabolism (2017)
-
BMP2 gene delivery to bone mesenchymal stem cell by chitosan-g-PEI nonviral vector
Nanoscale Research Letters (2015)
-
The role of transduced bone marrow cells overexpressing BMP-2 in healing critical-sized defects in a mouse femur
Gene Therapy (2015)
-
Healing of tibial and calvarial bone defect using Runx-2-transfected adipose stem cells
Tissue Engineering and Regenerative Medicine (2015)
-
Immobilization effect of bone morphogenetic protein-2 on collagen membrane via photoreactive gelatin derivatives: Biocompatibility and preservability of osteoinductive activity
Macromolecular Research (2015)