Abstract
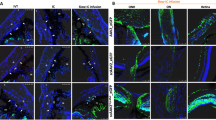
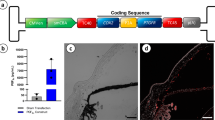
Obstruction of the aqueous humor outflow from the anterior chamber of the eye leads to an elevation of intraocular pressure in glaucoma, the second major cause of blindness worldwide. Our goal is to be able to modulate aqueous humor outflow resistance by gene transfer to the cells of the trabecular meshwork (TM). We have previously shown that adenoviral vectors are able to transfer a reporter gene to the TM of post-mortem human donors. However, assessing gene therapy for glaucoma requires models that can monitor changes in aqueous humor outflow facility (C = flow/pressure). In this study we used four replication-deficient adenoviruses in two such perfusion models. In the first model, whole porcine eyes were infected, perfused at constant pressure and flow changes recorded for 5 h. In the second one, anterior segments from human eyes were infected, perfused at constant flow and pressure changes recorded for 3 days. A single dose of 108 adenovirus plaque forming units (pfu) causes a reduction in C while single doses of 107, 106 and 105 p.f.u. do not affect outflow facility and retain positive gene transfer. These findings indicate that adenovirus, at effective doses, could become useful vectors for gene therapy of glaucoma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Quigley HA . Number of people with glaucoma worldwide (see comments) Br J Ophthalmol 1996 80: 389–393
Kupfer C, Underwood B, Gillen T . Leading causes of visual impairment worldwide In: Albert DM, Jakobiec FA (eds) . Principles and Practice of Ophthalmology Saunders: Philadelphia 1994 pp 1249–1255
Sommer A . Intraocular pressure and glaucoma Am J Ophthalmol 1989 107: 186–188
Quigley HA . Open-angle glaucoma (see comments) New Engl J Med 1993 328: 1097–1106
Rohen JW, van der E Zypen . The phagocytic activity of the trabecular meshwork endothelium. An electron-microscopic study of the vervet (Cercopithecus aethiops) Albrecht Von Graefes Archiv fur Klinische und Experimentelle Ophthalmologie 1968 175: 143–160
Bill A . The drainage of aqueous humor Invest Ophthalmol 1975 14: 1–3
Alexander JP, Samples JR, Van Buskirk EM, Acott TS . Expression of matrix metalloproteinases and inhibitor by human trabecular meshwork Invest Ophthalmol Vis Sci 1991 32: 172–180
Johnson DH, Richardson TM, Epstein DL . Trabecular meshwork recovery after phagocytic challenge Curr Eye Res 1989 8: 1121–1130
Quigley HA, Flower RW, Addicks EM, McLeod DS . The mechanism of optic nerve damage in experimental acute intraocular pressure elevation Invest Ophthalmol Vis Sci 1980 19: 505–517
Graham FL, Prevec L . Manipulation of adenoviral vectors In: Murray EJ (ed) . Methods in Molecular Biology Humana Press: Clifton, NJ 1991 pp 109–128
Berkner KL . Development of adenovirus vectors for the expression of heterologous genes Biotechniques 1988 6: 616–629
Rosenfeld MA et al. In vivo transfer of the human cystic fibrosis transmembrane conductance regulator gene to the airway epithelium Cell 1992 68: 143–155
Rosenfeld MA et al. Adenovirus-mediated transfer of a recombinant alpha 1-antitrypsin gene to the lung epithelium in vivo Science 1991 252: 431–434
Borrás T, Tamm ER, Zigler JS Jr . Ocular adenovirus gene transfer varies in efficiency and inflammatory response Invest Ophthalmol Vis Sci 1996 37: 1282–1293
Bennett J et al. Adenovirus vector-mediated in vivo gene transfer into adult murine retina Invest Ophthalmol Vis Sci 1994 35: 2535–2542
Budenz DL, Bennett J, Alonso L, Maguire A . In vivo gene transfer into murine corneal endothelial and trabecular meshwork cells Invest Ophthalmol Vis Sci 1995 36: 2211–2215
Abraham NG et al. Adenovirus-mediated heme oxygenase-1 gene transfer into rabbit ocular tissues Invest Ophthalmol Vis Sci 1995 36: 2202–2210
Fehervari Z et al. Gene transfer to ex vivo stored corneas Cornea 1997 16: 459–464
Larkin DF et al. Adenovirus-mediated gene delivery to the corneal endothelium Transplantation 1996 61: 363–370
Borrás T, Matsumoto Y, Epstein DL et al. Gene transfer to the human trabecular meshwork by anterior segment perfusion Invest Ophthalmol Vis Sci 1998 39: 1503–1507
Erickson-Lamy K, Schroeder AM, Bassett-Chu S, Epstein DL . Absence of time-dependent facility increase (‘washout’) in the perfused enucleated human eye Invest Ophthalmol Vis Sci 1990 31: 2384CC2388
Epstein DL et al. Experimental obstruction to aqueous outflow by pigment particles in living monkeys Invest Ophthalmol Vis Sci 1986 27: 387–395
Trapnell BC . Adenoviral vectors for gene transfer Adv Drug Del Rev 1993 12: 185–199
Erzurum SC et al. Protection of human endothelial cells from oxidant injury by adenovirus-mediated transfer of the human catalase cDNA Nucleic Acids Research 1993 21: 1607–1612
Wickham TJ, Mathias P, Cheresh DA, Nemerow GR . Integrins alpha v beta 3 and alpha v beta 5 promote adenovirus internalization but not virus attachment Cell 1993 73: 309–319
Wickham TJ, Filardo EJ, Cheresh DA, Nemerow GR . Integrin alpha v beta 5 selectively promotes adenovirus-mediated cell membrane permeabilization J Cell Biol 1994 127: 257–264
Mathias P, Wickham T, Moore M, Nemerow G . Multiple adenovirus serotypes use alpha v integrins for infection J Virol 1994 68: 6811–6814
Nevins JR et al. Regulation of the primary expression of the early adenovirus transcription units J Virol 1979 32: 727–733
Ellingsen BA, Grant WM . Trabeculotomy and sinusotomy in enucleated human eyes Invest Ophthalmol 1972 11: 21–28
Grant WM . Experimental aqueous perfusion in enucleated human eyes Arch Ophthalmol 1963 69: 783–801
Grant WM . Further studies on facility of flow through the trabecular meshwork Arch Ophthalmol 1958 60: 523–533
Rosenquist R et al. Outflow resistance of enucleated human eyes at two different perfusion pressures and different extents of trabeculotomy Curr Eye Res 1989 8: 1233–1240
Mäepea O, Bill A . Pressures in the juxtacanalicular tissue and Schlemm’s canal in monkeys Exp Eye Res 1992 54: 879–883
Grant WM, Schuman JS . The angle of the aanterior chamber In: Epstein DL, Allingham RR, Schuman JS (eds) . Chandler and Grant’s Glaucoma Williams & Wilkins, Baltimore, MD, 1996 pp 51–83
Oral HB et al. Ex vivo adenovirus-mediated gene transfer and immunomodulatory protein production in human cornea Gene Therapy 1997 4: 639–647
Huggert A . Pore size in the filtration angle of the eye Acta Ophthalmol 1955 33: 271–284
Huggert A . An experiment in determining the pore size distribution curve to the filtration angle of the eye Acta Ophthalmol 1957 35: 12–19
Barany E . Pore size and passage of particulate matter through the trabecular meshwork Doc Ophthalmol 1959 13: 41–55
Johnson M et al. The filtration characteristics of the aqueous outflow system Exp Eye Res 1990 50: 407–418
Stone EM et al. Identification of a gene that causes primary open angle glaucoma (see comments) Science 1997 275: 668–670
Masuda I, Matsuo T, Yasuda T, Matsuo N . Gene transfer with liposomes to the intraocular tissues by different routes of administration Invest Ophthalmol Vis Sci 1996 37: 1914–1920
Zabner J et al. Adenovirus-mediated gene transfer transiently corrects the chloride transport defect in nasal epithelia of patients with cystic fibrosis Cell 1993 75: 207–216
Channon KM et al. Adenoviral gene transfer of nitric oxide synthase: high level expression in human vascular cells Cardiovasc Res 1996 32: 962–972
Jones N, Shenk T . Isolation of adenovirus type 5 host range deletion mutants defective for transformation of rat embryo cells Cell 1979 17: 683–689
Friedman JM, Babiss LE, Clayton DF, Darnell JE, Jr . Cellular promoters incorporated into the adenovirus genome: cell specificity of albumin and immunoglobulin expression Molec Cell Biol 1986 6: 3791–3797
Graham FL, van der Eb AJ . A new technique for the assay of infectivity of human adenovirus 5 DNA Virology 1973 52: 456–467
Johnson DH, Tschumper RC . Human trabecular meshwork organ culture. A new method Invest Ophthalmol Vis Sci 1987 28: 945–953
Johnson DH, Tschumper RC . The effect of organ culture on human trabecular meshwork Exp Eye Res 1989 49: 113–127
Johnson DH . Human trabecular meshwork cell survival is dependent on perfusion rate Invest Ophthalmol Vis Sci 1996 37: 1204–1208
Epstein DL, Hashimoto JM, Anderson PJ, Grant WM . Effect of iodoacetamide perfusion on outflow facility and metabolism of the trabecular meshwork Invest Ophthalmol Vis Sci 1981 20: 625–631
Epstein DL et al. Influence of ethacrynic acid on outflow facility in the monkey and calf eye Invest Ophthalmol Vis Sci 1987 28: 2067–2075
Epstein DL, Hooshmand LB, Epstein MP . Thiol adducts of ethacrynic acid increase outflow facility in enucleated calf eyes Curr Eye Res 1992 11: 253–258
Epstein DL, Patterson MM, Rivers SC, Anderson PJ . N-ethylmaleimide increases the facility of aqueous outflow of excised monkey and calf eyes Invest Ophthalmol Vis Sci 1982 22: 752–756
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Borrás, T., Rowlette, L., Erzurum, S. et al. Adenoviral reporter gene transfer to the human trabecular meshwork does not alter aqueous humor outflow. Relevance for potential gene therapy of glaucoma. Gene Ther 6, 515–524 (1999). https://doi.org/10.1038/sj.gt.3300860
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3300860