Abstract
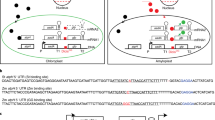
Most mitochondrial proteins are encoded by nuclear genes and are synthesized as precursors containing a presequence at the N terminus. In yeast and in mammalian cells, the function of the presequence in mitochondrial targeting has been revealed by chimaeric gene studies. Fusion of a mitochondrial presequence to a foreign protein coding sequence enables the protein to be imported into mitochondria in vitro as well as in vivo1–4. Whether plant mitochondrial presequences function in the same way has been unknown. We have previously isolated and characterized a nuclear gene (atp2–1) from Nicotiana plumbaginifolia that encodes the β-subunit of the mitochondrial ATP synthase5. We have constructed a chimaeric gene comprising a putative atp2–1 presequence fused to the bacterial chloramphenicol acetyltransferase (CAT) coding sequence and introduced it into the tobacco genome. We report here that a segment of 90 amino acids of the N terminus of the β-subunit precursor is sufficient for the specific targeting of the CAT protein to mitochondria in transgenic plants. Our results demonstrate a high specificity for organelle targeting in plant cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
1. Emr, S. et al. J. Cell Biol 102, 523–533 (1986). 2. Horwich, A., Kalousek, R, Mellman, I. & Rosenberg, I. EMBO J. 4, 1129–1135 (1985). 3. Hurt, E., Muller, U. & Schatz, G. EMBO J. 4, 3509–3518 (1985). 4. Van Loon, A. P., Brandli, A. W. & Schatz, G. Cell 44, 801–812 (1986). 5. Boutry, M. & Chua, N.–H. EMBO J. 4, 2159–2165 (1985). 6. Odell, J. T., Nagy, F. & Chua, N.–H. Nature 313, 810–812 (1985). 7. Morelli, G., Nagy, F., Fraley, R. T., Rogers, S. G. & Chua, N.–H. Nature 315,200–204 (1985). 8. Horsch, R. B. & Klee, H. J. Proc. natn. Acad. Sci. U.S.A. 83, 4428–4432 (1986). 9. Ochoa, S. Meth. Enzym. 1, 735–739 (1955). 10. Shaw, W. V. CRC crit. Rev. Biochem. 14, 1–46 (1982). 11. Hurt, E., Soltanifar, N., Goldschmidt–Clermont, M., Rochaix, J.–D. & Schatz, G. EMBO J. 4, 1343–1350 (1986). 12. Van den Broeck, G. et al. Nature 313, 358–363 (1985). 13. Schreier, P., Seftor, E. A., Schell, J. & Bohnert, H. J. EMBO J. 5, 1335–1342 (1985). 14. Poulsen, C., Fluhr, R., Kauffman, J. M., Boutry, M. & Chua, N.–H. Molec. gen. Genet. 205, 193–200 (1986). 15. Stueber, D., Ibrahimi, I., Cutler, D., Dobberstein, B. & Bujart, H. EMBO J. 3, 3143–3148 (1984). 16. Dente, L., Cesareni, G. & Cortese, R. Nucleic Acids Res. 11, 1645–1655 (1983). 17. German, C. M., Moffat, L. F. & Howard, B. H. Molec. cell. Biol 2, 1044–1051 (1982). 18. Suissa, M. Analyt. Biochem. 133, 511–514 (1983). 19. Hunt, A., Chu, N., Odell, J., Nagy, F. & Chua, N.–H. PL Molec. Biol. 8, 23–35 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Boutry, M., Nagy, F., Poulsen, C. et al. Targeting of bacterial chloramphenicol acetyltransferase to mitochondria in transgenic plants. Nature 328, 340–342 (1987). https://doi.org/10.1038/328340a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/328340a0
This article is cited by
-
Diversity in subcellular targeting of the PP2A B′η subfamily members
Planta (2009)
-
AtSufE is an essential activator of plastidic and mitochondrial desulfurases in Arabidopsis
The EMBO Journal (2006)
-
Molecular cloning and expression analyses of mitochondrial and plastidic isoforms of cysteine synthase (O-acetylserine(thiol)lyase) fromArabidopsis thaliana
Amino Acids (1999)
-
Mitochondrial and chloroplast targeting sequences in tandem modify protein import specificity in plant organelles
Plant Molecular Biology (1996)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.