Abstract
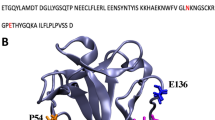
The epidermal growth factors (EGFs)1–3are powerful mitogens for a wide variety of cells in culture4; human EGF (hEGF), known as urogastrone, also inhibits gastric acid secretion in vivo1. The transforming growth factors (TGF-α) are related to the EGF family both in sequence5 and activity6,7and EGF-like sequences are often observed in a wide range of functionally unrelated proteins8. Attempts to examine the structure of EGF by diffraction methods have not yet succeeded because of difficulties with crystallization. We report here a three-dimensional structure of a biologically active derivative (residues 1–48) of the 53-residue human EGF. An analysis of high resolution 1H nuclear magnetic resonance (NMR) spectra was used together with a combination of distance geometry, restrained energy minimization and restrained molecular dynamics methods. The three-dimensional structure provides a basis for understanding the properties of EGFs and for predicting the structures of homologous sequences in other proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
1. Gregory, H. Nature 257, 325–327 (1975). 2. Cohen, S. /. bioL Chem. 237, 1555–1562 (1962). 3. Simpson, R. J. et al Bur. J. Biochem. 153, 629–637 (1985). 4. Schlessinger, J. et al CRC Crit. Rev. Biochem. 14, 93–111 (1983). 5. Marquardt, H. et al Proc. natn. Acad. Scl U.S.A. 80, 4684–4688 (1983). 6. Marquardt, H. & Todaro, G. J. /. biol Chem. 257, 5220–5225 (1982). 7. Anzano, M. A. et al Cancer Res. 42, 4776–4778 (1982). 8. Doolittle, R. F. Trends biochem. Sci 10, 233–237 (1985). 9. Carver, J. A. et al FEBS Letts 205, 77–81 (1986). 10. Montelione, G. T., Wiithrich, K., Nice, E. C., Burgess, A. W. & Scheraga, H. A. Proc. natn. Acad. Sci. U.S.A. 83, 8594–8598 (1986). 11. Williamson, M. P., Havel, T. F. & Wiithrich, K. /. molec. Biol 182, 295–315 (1985). 12. Kaptein, R., Zuiderweg, E. R. P., Scheek, R. M., Boelens, R. & van Gunsteren, W. F. /. molec. Biol 182, 179–182 (1985). 13. Crippen, G. M. & Havel, T. Ada Crystallogr. sect. A 34, 282–284 (1978). 14. van Gunsteren, W. F. & Berendsen, H. J. C. Biochem. Soc. Trans. 10, 301–305 (1982). 15. Mayo, K. H., Schaudies, P., Savage, C. R., De Marco, A. & Kaptein, R. Biochem. J. 239, 13–18 (1986). 16. Hollenberg, M. D. & Gregory, H. Molec. PharmacoL 17, 314–320 (1980). 17. Komoriya, A. et al Proc. natn. Acad. Scl U.S.A. 81, 1351–1355 (1984). 18. Holladay, L. A., Savage, C. R., Cohen, S. & Puett, D. Biochemistry 15, 2624–2633 (1976). 19. Dayhoff, M. O. Atlas of Protein Sequence and Structure Vol. 5, suppl. 3, p. 85 (Natn. Biomed. Res. Found., Washington, 1978). 20. Giinzler, W. A. et al Hoppe–Seyler's Z. physioL Chem. 363, 1155–1165 (1982). 21. McMullen, B. A. et al Biochemistry 22, 2875–2884 (1983). 22. Esmon, N. L., DeBault, L. E. & Esmon, C. T. /. biol Chem. 258, 5548–5553 (1983). 23. Rees, D. J. G. D. Phil thesis, University of Oxford (1986). 24. Stenflo, J., Lundwall, A & Dahlback, B. Proc. natn. Acad. Sci U.S.A. 84, 368–372 (1987). 25. Kurachi, K. & Davie, E. W. Proc. natn. Acad. Sci U.S.A 79, 6461–6464 (1982). 26. McMuilen, B. A., Fujikawa, K. & Kisiel, W. Biochem. biophys. Res. Comm. 115,8–14 (1983). 27. Fernlund, P. & Stenflo, J. /. biol Chem. 257, 12170–12179 (1982). 28. Lundwall, A et al Proc. natn. Acad. Sci U.S.A. 83, 6716–6720 (1986). 29. Arlaud, G. J., Willis, A. C. & Gagnon, J. Biochem. J. 241, 711–720 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cooke, R., Wilkinson, A., Baron, M. et al. The solution structure of human epidermal growth factor. Nature 327, 339–341 (1987). https://doi.org/10.1038/327339a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/327339a0
This article is cited by
-
Economic optimization of expression of soluble human epidermal growth factor in Escherichia coli
Biotechnology Letters (2022)
-
Aspartate/asparagine-β-hydroxylase crystal structures reveal an unexpected epidermal growth factor-like domain substrate disulfide pattern
Nature Communications (2019)
-
Structural basis for specific self-incompatibility response in Brassica
Cell Research (2016)
-
Iain D Campbell 1941–2014
Nature Structural & Molecular Biology (2014)
-
Expression line approach to recombinant human epidermal growth factor into the yeast, Pichia pastoris from Huh-7 cell line
Molecular Biology Reports (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.