Abstract
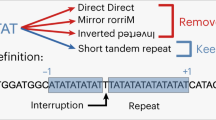
DNA regions which are composed of a single or relatively few short sequence motifs usually in tandem (‘pure simple sequences’) have been reported in the genomes of diverse species (for reviews see refs 1–4), and have been implicated in a range of functions including gene regulation (for reviews see refs 5–7), signals for gene conversion and recombination8–10, and the replication of telomeres11They are thought to accumulate by DNA slippage12–16 and mispairing during replication and recombination or extension of single-strand ends2,4,10,11. In order to systematize the range of DNA simplicity and the genetic nature of the regions that are simple, we have undertaken an extensive computer search of the DNA sequence library of the European Molecular Biology Laboratory (EMBL)17. We show here that nearly all possible simple motifs occur 5–10 times more frequently than equivalent random motifs. Furthermore, a new computer algorithm reveals the widespread occurrence of significantly high levels of a new type of ‘cryptic simplicity’ in both coding and noncoding DNA. Cryptically simple regions are biased in nucleotide composition and consist of scrambled arrangements of repetitive motifs which differ within and between species. The universal existence of DNA simplicity from monotonous arrays of single motifs to variable permutations of relatively short-lived motifs suggests that ubiquitous slippage-like mechanisms are a major source of genetic variation in all regions of the genome, not predictable by the classical mutation process.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Hamada, H., Petrino, M. G. & Kakunaga, T. Proc. natn. Acad. Sci. U.S.A. 79, 6465–6469 (1982).
Tautz, D. & Renz, M. Nucleic Acids Res. 12, 4127–4138 (1984).
Greaves, D. R. & Patient, R. K. EMBO J. 4, 2617–2626 (1985).
Dover, G. A. & Tautz, D. Phil. Trans. R. Soc. 312, 275–290 (1986).
Wang, A. J. H. et al. Nature 282, 680–686 (1979).
Weintraub, H. & Groudine, M. Science 193, 348–856 (1976).
Hentschel, C. C. Nature 295, 714–716 (1982).
Shen, S. H., Slighton, J. L. & Smithies, O. Cell 26, 191–203 (1981).
Goodman, M. Bioessays 3, 9–14 (1985).
Jeffreys, A. J., Wilson, V. & Thein, S. L. Nature 314, 67–72 (1985).
Blackburn, E. & Szostak, J. A. Rev. Biochem. 53, 163–152 (1984).
Drake, J. W., Glickman, B. W. & Ripley, L. S. Am. Scient. 71, 621–630 (1983).
Streisinger, G. et al. Cold Spring Harb. Symp. quant. Biol. 31, 77–84 (1966).
Wells, R. D., Ohtsuka, E. & Khorana, H. G. J. molec. Biol. 14, 221–240 (1965).
Morgan, A. R. et al. Biochemistry 13, 1596–1603 (1974).
Efstratiadis, A. et al. Cell 21, 653–668 (1980).
Cameron, G. et al. EMBL Sequence Data Library Release 4 (1985).
Bird, A. P. Nucleic Acids Res. 8, 1499–1504 (1980).
Straus, N. A. & Birnboim, H. C. Biochim. biophys. Acta 454, 419–428 (1976).
Karlin, S. & Ghandour, G. Proc. natn. Acad. Sci. U.S.A. 5800-5804 (1985).
Karlin, S., Ghandour, G. & Foulser, D. E. Molec. Biol. Evol. 2, 35–45 (1985).
Rogers, J. Nature 305, 101–102 (1983).
Yaffe, D. et al. Nucleic Acids Res. 13, 3723–3737 (1985).
Steinert, P. M. et al. Nature 302, 794–800 (1983).
Hassouna, N., Michot, B. & Bachellerie, J. P. Nucleic Acids Res. 12, 3563–3583 (1984).
Moore, G. P. Trends biochem. Sci. 8, 411–414 (1983).
Jones, W. C. & Kafatos, F. C. J. molec. Evol. 19, 87–103 (1982).
Brown, S. D. M. & Piechaczyk, M. J. molec. Biol. 165, 249–256 (1983).
Ohno, S. & Epplen, J. T. Proc. natn. Acad. Sci. U.S.A. 80, 3391–3395 (1983).
Dover, G. A. Nature 299, 1121–119 (1982).
Coen, E. S., Thoday, J. M. & Dover, G. A. Nature 295, 564–568 (1982).
Strachan, T., Webb, D. A. & Dover, G. A. EMBO J. 4, 1701–1708 (1985).
Dover, G. A. & Flavell, R. B. Cell 38, 623–624 (1984).
Ohta, T. & Dover, G. A. Genetics 108, 501–528 (1984).
Kneale, G. G. & Kennard, O. Biochem. Soc. Trans. 12, 1011–1014 (1984).
Kost, T. A. et al. Nucleic Acids Res. 11, 8287–8301 (1983).
Nudel, U. et al. Nucleic Acids Res. 11, 1759–1771 (1983).
Hanukoglu, I. & Fuchs, E. Cell 33, 915–924 (1983).
Hadjiolov, A. A. et al. Nucleic Acids Res. 12, 3677–3583 (1984).
Chan, Y. L., Olvera, J. & Wool, I. G. Nucleic Acids Res. 11, 7819–7831 (1983).
Dover, G. A. Trends Genet. 2, 159–165 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tautz, D., Trick, M. & Dover, G. Cryptic simplicity in DNA is a major source of genetic variation. Nature 322, 652–656 (1986). https://doi.org/10.1038/322652a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/322652a0
This article is cited by
-
Large-scale analysis reveals that the genome features of simple sequence repeats are generally conserved at the family level in insects
BMC Genomics (2017)
-
Application of molecular markers in plant genome analysis: a review
The Nucleus (2017)
-
Genome-wide mining, characterization, and development of microsatellite markers in Marsupenaeus japonicus by genome survey sequencing
Chinese Journal of Oceanology and Limnology (2017)
-
Genome microsatellite diversity within the Apicomplexa phylum
Molecular Genetics and Genomics (2016)
-
Nucleolar DNA: the host and the guests
Histochemistry and Cell Biology (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.