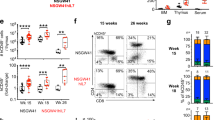
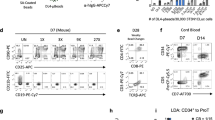
Abstract
There is much interest in early T-cell development, particularly in relation to the diversification of the T-cell receptor repertoire1,2 and the elucidation of the lineage relationships between T-cell populations in the thymus and peripheral lymphoid organs (reviewed in refs 3, 4). However, the requirements for the growth of the earliest thymic T-cell precursor in 13–14-day mouse embryo thymus in isolation from the thymic environment are unknown. Proliferation and maturation of such cells are not sustained either in the presence of monolayers of thymic stromal cells (refs. 5, 6 and see below) or by the addition of interleukin-2 (IL-2), despite the expression of receptors for this growth factor on a proportion of thymocytes displaying the immature Thy 1+ Lyt-2−L3T4− phenotype in the embryonic thymus7,8. In contrast, when maintained within the intact thymic environment in organ cultures9,10, 13–14-day thymic stem cells do show a pattern of surface marker and functional development similar to that seen in vivo, suggesting that short-range growth signals, perhaps necessitating direct contact with organized epithelial cells, are required. We have shown, by exploiting the selective toxicity of deoxyguanosine (dGuo) for early T cells, that this organ culture system can be manipulated to produce alymphoid lobes that can be recolonized from a source of precursors in a transfilter system11. We now show that recolonization of alymphoid lobes can also be achieved by association with T-cell precursors in hanging drops, allowing recolonization by exposure to defined numbers of precursors, including a single micromanipulated stem cell. Analysis of T-cell marker expression in these cultures shows that a single thymic stem cell can produce progeny of distinct phenotypes, suggesting that these marker-defined populations are not derived from separate pre-thymic precursors, but arise within the thymus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Snodgrass, H. R., Kisielow, P., Kiefer, M., Steinmetz, M. & von Boehmer, H. Nature 313, 592–595 (1985).
Raulet, D. M., Garman, R. D., Saito, M. & Tonegawa, S. Nature 314, 103–107 (1985).
Mathieson, B. J. & Fowlkes, B. J. Immun. Rev. 82, 141–173 (1984).
Scolley, R., Bartlett, P. & Shortman, K. Immun. Rev. 82, 79–103 (1984).
Jordan, R. K., Crouse, D. A. & Owen, J. J. T. J. reticuloendothel. Soc. 26, 385–399 (1979).
Jones, K. H. & St Peirre, R. L. In Vitro 17, 431–440 (1981).
Ceredig, R., Lowenthal, J. W., Nabholz, M. & MacDonald, H. R. Nature 314, 98–100 (1985).
Raulet, D. M. Nature 314, 101–103 (1985).
van Ewijk, W., Jenkinson, E. J. & Owen, J. J. T. Eur. J. Immun. 12, 262–271 (1982).
Kisielow, P., Lieserson, W. & von Boehmer, H. J. Immunology 133, 1117–1123 (1984).
Jenkinson, E. J., Franchi, L., Kingston, R. & Owen, J. J. T. Eur. J. Immun. 12, 583–587 (1982).
Ceredig, R., Dialynas, D. P., Fitch, F. W. & MacDonald, H. R. J. exp. Med. 158, 1654–1671 (1983).
Fontaine-Perus, J. C., Calman, F. M., Kaplan, C. & LeDouarin, N. M. J. Immun. 126, 2310–2316 (1981).
Wallis, V. J., Leuchars, E., Chwalinski, S. & Davies, A. J. S. Transplantation 19, 2–11 (1975).
Enzine, S., Weissman, I. L. & Rowse, R. V. Nature 309, 629–631 (1984).
Sharrow, S., Singer, A., Hammerling, U. & Mathieson, B. J. Transplantation 35, 355–362 (1983).
Lepault, F., Coffman, R. L. & Weissman, I. L. J. Immun. 131, 64–69 (1983).
Jotereau, F. V. & LeDouarin, N. M. J. Immun. 129, 1869–1877 (1982).
Micklem, H. S. et al. Transplantation 6, 299–302 (1968).
Bradley, S. M., Morrissey, P. M., Sharrow, S. O. & Singer, A. J. exp. Med. 155, 1638–1652 (1982).
Chervenak, R., Moorhead, J. W. & Cohen, J. J. J. Immun. 134, 695–698 (1985).
Pierres, A. et al. J. Immun. 132, 2775–2782 (1984).
Kingston, R., Jenkinson, E. J. & Owen, J. J. T. Eur. J. Immun. 14, 1052–1056 (1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kingston, R., Jenkinson, E. & Owen, J. A single stem cell can recolonize an embryonic thymus, producing phenotypically distinct T-cell populations. Nature 317, 811–813 (1985). https://doi.org/10.1038/317811a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/317811a0
This article is cited by
-
The development and function of thymic B cells
Cellular and Molecular Life Sciences (2015)
-
Detection of a CD4+CD8−CD3− cell subpopulation during the differentiation of cord blood CD34+ cells into T cells in vitro
Archivum Immunologiae et Therapiae Experimentalis (2009)
-
Development of αβ T cells in the human thymus
Nature Reviews Immunology (2002)
-
Spontaneous generation of human CD8+ TCRαβ+ cells derived from precursors within the double negative compartment
Immunology & Cell Biology (1995)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.