Abstract
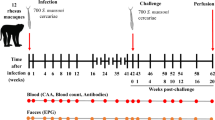
Studies of host resistance to parasite infection are usually based on experimental designs involving a primary infection and subsequent challenge exposure, resistance being recorded as the percentage reduction in parasite establishment in challenged hosts when compared with that in uninfected animals1–8. Few studies have focused on the dynamic nature of helminth establishment and mortality (and their presumed dependency on the rate of current exposure and past experiences of infection) in hosts repeatedly exposed to low levels of infection9–11. Here, we report the results of population studies on the dynamics of resistance to Schistosoma mansoni infection (a helminth parasite) in mice repeatedly exposed to cercarial invasion. Parasite burdens created by different levels and durations of exposure to infection reflect a dynamic interplay between rates of helminth establishment and mortality. Depending on the intensity of exposure, changes in worm load with duration of host infection vary from monotonie growth to a stable average parasite burden to convex curves in which the average load attains a maximum value before decaying in old animals. These trends are similar to observed patterns of S. mansoni infection in human communities12–14.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Wakelin, D. Immunity to Parasites (Arnold, London, 1984).
Butterworth, A. E., Taylor, D. W. & Veith, M. C. Immun. Rev. 61, 5–39 (1982).
Cohen, S. & Warren, K. S. Immunology of Parasitic Infections (Black well Scientific, Oxford, 1982).
Mitchell, G. F. Adv. Immun. 28, 451–467 (1980).
Anderson, R. M. & May, R. M. Adv. Parasit. 24, 1–101 (1985).
Taylor, A. E. R. & Muller, R. (eds) Br. Soc. Parasit. Symp. 18 (Blackwell Scientific, Oxford, 1980).
Sturrock, R. F. et al. Trans. R. Soc. trop. Med. Hyg. 75, 219–231 (1981).
Dean, D. A. Expl Parasit. 55, 1–104 (1983).
Jenkins, D. C. & Phillipson, R. F. Parasitology 62, 457–465 (1970).
Jenkins, D. C. & Phillipson, R. F. Int. J. Parasit. 2, 105–111 (1972).
Sturrock, R. F., Cottrel, B. J. & Kimani, R. Parasitology 88, 505–514 (1984).
Cheever, A. W., Kamel, J., Elwi, A. M., Musiman, J. E. & Danner, R. Am. J. trop. Med. Hyg. 26, 702–716 (1977).
Scott, D., Senker, K. & England, E. C. Bull Wld Hlth Org. 60, 89–100 (1982).
Bradley, D. J. & McCullough, F. Trans. R. Soc. trop. Med. Hyg. 67, 491–513 (1973).
Anderson, R. M. Parasitology 72, 281–305 (1976).
Warren, K. S. J. infect. Dis. 127, 595–609 (1973).
Dalton, P. R. & Pole, D. Bull Wld Hlth Org. 563, 417–424 (1978).
Andrade, Z. A. & Warren, K. S. Trans. R. Soc. trop. Med. Hyg. 58, 53–57 (1964).
Anderson, R. M. & May, R. M. Nature 315, 493–496 (1985).
Jordan, P. & Webbe, G. Schistosomiasis: Epidemiology, Treatment and Control (Heinemann, London, 1982).
Gonnert, R. & Andrews, P. Z. Parasit. 52, 129–150 (1977).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Crombie, J., Anderson, R. Population dynamics of Schistosoma mansoni in mice repeatedly exposed to infection. Nature 315, 491–493 (1985). https://doi.org/10.1038/315491a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/315491a0
This article is cited by
-
What is required in terms of mass drug administration to interrupt the transmission of schistosome parasites in regions of endemic infection?
Parasites & Vectors (2015)
-
Acquired immunity and epidemiology of Schistosoma haematobium
Nature (1991)
-
On the solution of mathematical models of herd immunity in human helminth infections
Journal of Mathematical Biology (1989)
-
Herd immunity to helminth infection and implications for parasite control
Nature (1985)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.