Abstract
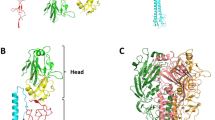
Antigenic variation in the haemagglutinin (HA) glycoprotein of influenza virus is associated with recurrent epidemics of respiratory disease in man (for review see ref. 1). We have examined the size of structural changes necessary to alter the antigenicity of HA by determining the three-dimensional structure of the HA from an antigenic mutant containing a single amino acid substitution which was selected by growth of virus in the presence of monoclonal antibodies. Here we present evidence that the simple addition of an amino acid side chain which results in only minor local distortions of the structure of the HA is sufficient structural alteration for a virus to escape neutralization by a monoclonal antibody. Our results also demonstrate that single amino acid substitutions can cause only local changes in the HA structure, verifying the assumption made in several studies to locate antigenic sites on the HA2–5 and other molecules6,7 and indicate that proposals8,9 of large conformational changes to account for variations in HA antigenicity are unnecessary in this case. The structure of the variant antigen has independently been successfully predicted (M. Karplus, personal communication).
Similar content being viewed by others
Article PDF
References
Webster, R. G. & Laver, W. G. in Influenza Viruses and Influenza (ed. Kilbourne, E. D.) 270–314 (Academic, New York, 1975).
Wiley, D. C., Wilson, I. A. & Skehel, J. J. Nature 289, 373–378 (1981).
Caton, J. J., Brownlee, C. C., Yewdell, C. A. & Gerhard, W. Cell 31, 417–427 (1982).
Daniels, R. S., Douglas, A. R., Skehel, J. J. & Wiley, D. C. J. gen. Virol. 64, 1657–1662 (1983).
Daniels, R. S. et al. in The Origin of Pandemic Influenza Viruses (eds Laver, G. W. & Chu, C. M.) (Elsevier, New York, in the press).
Colman, P. M., Varghese, J. N. & Laver, W. D. Nature 303, 41–44 (1983).
Evans, D. M. A., Minor, P. D., Schild, G. S. & Almond, J. W. Nature 304, 459–462 (1983).
Laver, W. G., Air, G. M. & Webster, R. G. in Genetic Variation Among Influenza Viruses (ed. Nayak, D. P.) 283–295 (Academic, New York, 1981).
Webster, R. G. & Laver, W. G. Virology 104, 139–148 (1980).
Bull. Wld Hlth Org. 45, 119–124 (1971).
Laver, W. G., Air, G. M., Dophiede, T. A. & Ward, C. W. Nature 283, 454–457 (1980).
Skehel, J. J. et al. Proc. natn. Acad. Sci. U.S.A. (in the press).
Rogers, G. N. et al. Nature 304, 76–78 (1983).
Wiley, D. C. & Skehel, J. J. J. molec. Biol. 112, 343–347 (1977).
Wilson, I. A., Skehel, J. J. & Wiley, D. C. Nature 289, 368–373 (1981).
Winkler, F. K., Schutt, C. E. & Harrison, S. C. Acta crystallogr. A 35, 901–911 (1979).
Sanger, F., Nicklen, S. & Coulson, A. R. Proc. natn. Acad. Sci. U.S.A. 74, 5463–5467 (1977).
Patel, T. P. et al. Nucleic Acids Res. 10, 5605–5620 (1982).
Verhoeyen, M. et al. Nature 286, 771–776 (1980).
Diamond, R. in Biomolecular Structure, Conformation, Function and Evolution Vol. 1 (ed. Srinivasan, R.) 567–588 (Pergamon, Oxford, 1980).
Hendrickson, W. A. & Konnert, J. in Biomolecular Structure, Function, Conformation, and Evolution Vol. 1 (ed. Srinivasan, R.) 43–47 (Pergamon, Oxford, 1980).
Cochran, W. Acta crystallogr. 4, 408–411 (1951).
Brooks, B. et al. J. comp. Chem. 4, 187–217 (1983).
Gelin, B. R. & Karplus, M. Biochemistry 18, 1256–1268 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Knossow, M., Daniels, R., Douglas, A. et al. Three-dimensional structure of an antigenic mutant of the influenza virus haemagglutinin. Nature 311, 678–680 (1984). https://doi.org/10.1038/311678a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/311678a0
This article is cited by
-
Antigen distortion allows influenza virus to escape neutralization
Nature Structural Biology (1998)
-
Structure of influenza virus haemagglutinin complexed with a neutralizing antibody
Nature (1995)
-
Multiple amino acid substitutions in the HN protein of the paramyxovirus, SV 5, are selected for in monoclonal antibody resistant mutants
Archives of Virology (1993)
-
Structural basis of antigenic variation: Studies of influenza virus neuraminidase
Immunology & Cell Biology (1992)
-
Three antibody molecules can bind simultaneously to each monomer of the tetramer of influenza virus neuraminidase and the trimer of influenza virus hemagglutinin
Archives of Virology (1991)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.