Abstract
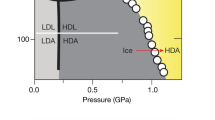
Amorphous solids are made mainly by cooling the liquid below the glass transition without crystallizing it, a method used since before recorded history1, and by depositing the vapour onto a cold plate2, as well as by several other methods3,4. We report here a new way—by ‘melting’ a solid by pressure below the glass transition of the liquid—and apply it to making a new kind of amorphous ice. Thus, ice I has been transformed to an amorphous phase, as determined by X-ray diffraction, by pressurizing it at 77 K to its extrapolated melting point of 10 kbar. At the melting point, the fluid is well below its glass transition. On heating at a rate of ∼2.6 K min−1 at zero pressure it transforms at ∼117 K to a second amorphous phase with a heat evolution of 42±∼8 J g−1 and at ∼152 K further transforms to ice I with a heat evolution of 92±∼15 J g−1. In one sample, ice Ic was formed and in another, existing crystals of ice Ih grew from the amorphous phase. Heating below the 117 K transition causes irreversible changes in the diffraction pattern, and a continuous range of amorphous phases can be made. Similar transformations will probably occur in all solids whose melting point decreases with increasing pressure if they can be cooled sufficiently for a transformation to a crystalline solid to be too slow.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Morey, A. W. The Properties of Glass 2nd edn, Ch. l (Reinhold, New York, 1954).
Tammann, G. & Starinkewitsch, J. Z. phys. Chem. 85, 573–578 (1913).
Secrist, D. R. & McKenzie, J. D. in Modem Aspects of the Vitreous State Vol. 3, 149–165 (Butterworths, London, 1964).
De Carli, P. S. & Jamieson, J. C. J. chem. Phys. 31, 1675–1676 (1959).
Bertie, J. E., Calvert, L. D. & Whalley, E. J. chem. Phys. 38, 840–846 (1963).
Burton, E. F. & Oliver, W. F. Proc. R. Soc. A153, 166–172 (1938).
Dowell, L. G. & Rinfret, A. P. Nature 188, 1144–1148 (1960).
Bondot, P. C.r. hebd. Séanc. Acad. Sci. Paris 265, 316–318 (1967).
Narten, A. H., Venkatesh, C. G. & Rice, S. A. J. chem. Phys. 64, 1106–1121 (1976).
Mayer, E. & Brügeller, P. Nature 298, 715–718 (1982).
Ghormley, J. A. & Hochandel, C. J. Science 171, 62–64 (1971).
Gough, R. & Davidson, D. W. Can. J. Chem. 49, 2691–2699 (1971).
Kuriakose, A. K. & Whalley, E. J. Chem. Phys. 48, 2025–2031 (1968).
Merrill, Leo J. phys. Chem. Ref. Data 6, 1205–1252 (1977).
Cannon, J. F. J. phys. Chem. Ref. Data 3, 798–824 (1974).
Stillinger, F. H. & Schweitzer, K. S. J. phys. Chem. 87, 4281–4288 (1983).
Bridgman, P. W. Proc. Am. Acad. Arts Sci. 76, 9–24 (1945).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mishima, O., Calvert, L. & Whalley, E. ‘Melting ice’ I at 77 K and 10 kbar: a new method of making amorphous solids. Nature 310, 393–395 (1984). https://doi.org/10.1038/310393a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/310393a0
This article is cited by
-
Liquid-liquid phase separation in supercooled water from ultrafast heating of low-density amorphous ice
Nature Communications (2023)
-
Tracking cubic ice at molecular resolution
Nature (2023)
-
Evidence for a rosiaite-structured high-pressure silica phase and its relation to lamellar amorphization in quartz
Nature Communications (2023)
-
Molecular dynamics analysis of elastic properties and new phase formation during amorphous ices transformations
Scientific Reports (2022)
-
Evidence of a liquid–liquid phase transition in H\(_2\)O and D\(_2\)O from path-integral molecular dynamics simulations
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.