Abstract
Study design:
Case report.
Summary of background data:
Traumatic atlanto-occipital dislocation (AOD) with a complete medulla/spinal cord transection is rare and believed to be fatal owing to the high level of the spinal cord injury. Clinical outcome is poor. Consequently, relatively few case reports of adult patients surviving this injury appear in the literature.
Objectives and result:
We present the case of a 20-year-old male, who sustained an AOD with a complete medulla/spinal cord transection in a motorcycle accident to discuss the possibility of long-term survival with this condition. The patient underwent occipito-cervical stabilization. With an Injury Severity Score of 75, by definition unsurvivable, the patient is 16 months after the injury, ventilated and fully dependent for all care.
Conclusion:
Long-term survival following AOD with a complete medulla/spinal cord transection is possible if immediate resuscitation at the scene is available. Nevertheless, it remains questionable whether or not a patient with such a devastating injury and without any prospect of functional recovery should be kept alive under all circumstances.
Similar content being viewed by others
Introduction
Traumatic atlanto-occipital dislocation (AOD) is a devastating injury to the upper cervical spine and spinal cord, and involves complete disruption of all the ligaments between the occiput and the atlas (C1).1 AOD is the cause of death in an estimated 8–35% of traffic fatalities and 10% of fatal cervical spine injuries.2, 3 The first documented case of AOD with survival was described by Blackwood in 1908.4 Although there are over 200 patients documented to have survived AOD, only half have shown long-term survival, mainly being children and young adults.5, 6, 7, 8
The craniocervical junction is comprised of the middle-atlantoaxial joint, and the paired lateral atlantoaxial and atlanto-occipital articulations.9 Fractures and dislocations of the craniocervical junction represent one-third of all injuries to the cervical spine. They are usually caused by high-energy trauma such as traffic accidents or falls from significant heights.10, 11, 12, 13 Dislocations of the atlanto-occipital joint are classified into three groups: anterior, longitudinal and posterior injuries.14 The complete loss of articular continuity and functionality of the stabilizing structure render these injuries highly unstable. AOD is associated with injuries to the brainstem, high level spinal cord injuries and lesions to the vascular structures of the neck.15, 16, 17 Serious complications include unconsciousness, respiratory and cardiac arrest, subarachnoid haemorrhage, brainstem shear injury and fourth ventricle haematomas.12, 18, 19 Ultimately, surviving AOD patients have total neurological impairment from the atlanto-occipital joint down.
At patient presentation, identification of AOD, and subsequent therapeutic intervention, is hampered by the frequent combination of AOD with traumatic brain injury and/or polytrauma. AOD usually results in immediate death, although a small number of cases survive for a short time. Long-term survival is rare.18 Survival, in part, depends on prompt intervention, namely respiratory support and neurologic preservation, being careful immobilization and manoeuvring of the head and cervical spine.20 Although prompt recognition of AOD and stabilization is essential to prevent mortality and further morbidity, the general outlook for these patients is poor.9, 19, 21 In this paper, we present the first known case of a patient to survive AOD with complete transection at the highest level of the spinal cord.
Case report
A 20-year-old Caucasian male, without significant medical history, lost control of his motorcycle travelling at high speed. Bystanders observed that he became airborne and hit a tree branch, which made contact with his upper neck/chest region. The helmet he was wearing was dislodged. A physician at the scene ran to his aid and gave immediate mouth-to-mouth resuscitation. The patient was unconscious and had a Glasgow Coma Scale (GCS) score of 3 at the scene. The ambulance arrived and administered cervical spine and backboard precautions. Following successful resuscitation and application of a laryngeal mask, the patient was transferred sedated and relaxed to the emergency department of the Fremantle Hospital in Western Australia.
The patient arrived at hospital unresponsive with a heart rate of 66 b.p.m. and a blood pressure of 85/27 mmHg. The GCS was unchanged, and his pupillary light reactions showed pinpoint, equal and reactive pupils on both sides. He had normal heart sounds with a regular rate and rhythm. His extremities were warm and peripheral pulses were easily palpable. Motor responses were absent in the upper and lower limbs as well as the neck, indicative of complete quadriplegia. There was no spontaneous respiratory effort, even since the accident. The patient was noted to be blinking his left eyelid, but apparently not to command. The Trauma Score was 6/16 on arrival in the emergency department.22 Philadelphia collar was applied and remained in position throughout the subsequent investigations.
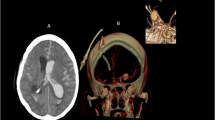
A plain radiograph of his cervical spine revealed a disarticulation of the atlas from the skull base, and the cervical spine was posteriorly displaced by about 2 cm in relation to the occipital condyles, which were no longer articulating with the cervical spine (Figure 1).
With the exception of a fracture line at the base of the skull and diffuse cerebral swelling, evidenced in the computed tomography (CT) scan, no skeletal pathological findings were identified from the clinical and radiological work-up. The CT of the cervical spine showed an atlanto-occipital disarticulation with posteriorly dislocated lateral masses of C1 in relation to the occipital condyles. No fracture was identified in the odontoid or at any other level of the cervical spine (Figure 2). There was obvious swelling on the anterior surface of the neck. A large anterior paravertebral soft tissue haematoma/swelling was showed. Other injuries included multiple facial abrasions and a periorbital haematoma and soft tissue injuries to limbs and swelling on the right side, but no apparent major chest/abdominal or pelvic trauma. Following these investigations, the patient was transferred to the Intensive Care Unit (ICU) and assessed by the spinal injury orthopaedic surgeon on call, being 4½ hours after admission.
On the following day, neurological examination showed no upper or lower limb movement, either spontaneously, to verbal command or to painful stimulus. His degree of cognitive awareness was difficult to ascertain. After wakening the patient, he showed an apallic syndrome, similar to a persistent vegetative state, suggestive of a high cervical spine lesion. A magnetic resonance image (MRI) undertaken 2 days post-trauma confirmed the diagnosis of an AOD with a complete transection of the lower medulla and upper cervical cord. The cord margins were widely separated. An extensive prevertebral parapharyngeal haemorrhage was noted, as was oedema with extensive fluid signal and oedema related to the posterior elements and soft tissues between the base of skull and C1/2 (Figure 3).
In the following days, repeated neurological assessments revealed complete paralysis and sensory deficit below neck level. Extrinsic eye movements and pupillary reflexes were intact, as were the cranial nerves II to VIII inclusive. However, there was no evidence of functionality of IX, X, XI and XII. The gag reflex was absent. These neurological findings did not change during the remainder of his hospitalization. He was conscious and responding using eye signals. The patient blinked in response to questions: once for ‘yes’ and twice for ‘no’. Using this method of communication, it was established that he was not in any pain. His eye signs indicated a syndrome of the dorsal midbrain and evidence of a pontine injury. The attending neurologist subsequently diagnosed Locked-In syndrome (LIS). Six days after admission, the patient experienced complete lung collapse and consolidation on the left side, but this responded to PEEP-ventilation, physiotherapy and appropriate antibiotics. Owing to the patient's extreme impairment, tracheostomy and endoscopically assisted percutaneous gastroenterostomy were performed. Physiotherapists, occupational and speech therapists were involved early in his treatment. He learnt to attract attention by lip pursing and exhaling a small volume of air. The patient had a clinically difficult course in ICU. He overcame a variety of recurrent infections, including ventilator-acquired pneumonia, UTIs, bacterial sinusitis and frequent fever of unknown origin. Recurrent lower respiratory tract infections with Klebsiella oxytoca and Staphylococcus aureus were treated with Ticarcillin.
After 7 weeks in ICU at Fremantle Hospital, the patient was transferred to Royal Perth Hospital, the level 1 trauma center of Western Australia. He then underwent occipito-cervical stabilization surgery.
Under general anaesthesia, prone position using Mayfield skull callipers, a posterior occipito-cervical exposure was performed. Owing to the wide separation of the occipito–cervical junction and high risk of vascular damage, with potential of severe cerebral compromise, the operation was performed without reduction to the anatomical position. Using a multisegmental screw/rod titanium construct, very secure fixation from the occiput to the C4 vertebra was achieved (Figure 4). The fixation was supplemented by iliac crest graft. The surgery was without complication and no postoperative orthosis was necessary. Fifty days post-surgery, the patient was transferred to the Royal Perth Hospital's rehabilitation campus. Sixteen months after the accident, the patient is still undergoing rehabilitation. For 2 hours daily, he is allowed out of bed in a custom-made wheel chair with portable ventilation, but otherwise he is dependent for all care.
Discussion
We have reported the unique case of a patient who survived a posterior AOD with complete transection of the spinal cord. Traumatic AOD commonly results in death.23 It is thought that the pathomechanism involves flexion and distraction, as well as rotation and hyperextension.24, 25 Rupture of the alar ligaments and the tectorial membrane, as well as the occipito-atlantal joint capsules and membranes, lead to severe C0-C1 instability in a vertical, anterior or posterior direction.26, 27 Diagnosis is usually established from plain lateral cervical radiographs in most cases,28, 29 but CT scan with sagittal reconstruction or MRI may be required when diagnosis is questionable.9, 30 In the case of our patient, the AOD was clearly evident in the plain cervical spine radiograph (Figure 1). Additional MRI confirmed the diagnosis and clarified adjacent soft tissue injuries and spinal cord involvement (Figure 3). Patients with associated lesions of the carotid or vertebral arteries are at a high risk of mortality,31, 32 in which case standard four-vessel angiography is advocated to further investigate the vascular supply. Although we did not perform an angiography in our case, a disruption of the vascular supply may have caused the large haematoma.
As dislocation at this level can cause injury to the brainstem, cranial nerves, upper spinal cord and cervical roots, survivors usually present with a variety of symptoms and signs.5, 8 Respiratory insufficiency and cardiac arrhythmia occur frequently and, together with respiratory distress, may be secondary to brainstem compression, or lower cranial nerve palsies causing airway obstruction, in addition to diaphragmatic paralysis.29, 33 Neck swelling can also be an important sign of AOD in a coma patient.34, 35 Hypertension can occur owing to bilateral glossopharyngeal nerve lesions,36 whereas complete cord transection can cause hypotension.37 Medullary compression can lead to bradycardia and apneic spells.38 Other brainstem signs commonly observed with AOD include rotatory nystagmus, ocular bobbing and decerebrate posturing.32
AOD management focuses mainly on maintaining adequate ventilation and stabilization of the atlanto-occipital joint.39 It is based on several key principles. First and foremost is strict adherence to advanced trauma and life support principles: life-threatening injuries should receive priority. Next is stabilization, maintenance of alignment and definitive fixation.40, 41 Spinal fusion is the preferred method of treating AOD. It aims to achieve joint stability, thus preventing further damage to the spinal cord or brainstem, which is paramount in cases of incomplete cord injury.20 Death is likely within the first few weeks after initial survival; therefore, early stabilization surgery is often delayed, as was the case in our patient. The goals of treating AOD are spinal stability, neurologic preservation and to provide the best environment that will maximize neurological recovery.1, 42 Our patient underwent surgical stabilization, albeit delayed, from the occiput to the C4 vertebra. Stabilization beyond this point was thought to offer no further stability, but a more extensive instrumentation would possibly cause unnecessary rigidity that was likely to interfere with nursing the patient and lessen the patient's quality of life. Because it was obvious from the outset that our patient had no chance of any neurological recovery, early stabilization was not a high priority. Early stabilization was not a priority upon admission to hospital because it was deemed that the patient had nil chance of neurological recovery. Moreover, whether this patient survived beyond the first 2 weeks of admission was doubtful. However, in retrospect and in accord with the current literature, early stabilization, within the first 2 weeks of injury, could have improved long-term anatomical position and facilitated the nursing care of this patient.
With improvements in emergency transport services and advanced trauma life support, the number of patients with AOD admitted alive is increasing.6, 7, 35, 43 Standard radiographs of the cervical spine usually show the lesion. In cases where there has been spontaneous reposition of the dislocation, careful examination under an image intensifier is likely to show the region of instability. Conversely, with cases of persisting dislocation, immediate reposition and stabilization would be mandatory, if there is potential for neurological recovery. The role of traction is controversial. Traction may be helpful for achieving an anatomic correct position, but it may also be harmful.44, 45 However, in the case of AOD with longitudinal distraction, further traction is not indicated.
Our patient had a tragic accident that resulted in a devastating injury. He sustained a complete upper cervical spine cord/brainstem injury at the time of the motorbike crash. Interestingly, the patient had an Injury Severity Score (ISS) of 75, which is, by definition, an unsurvivable injury (complete cervical spinal cord injury of the C3 and above, with or without dislocation or fracture, has an Abbreviated Injury Score of 6, which results in an ISS of 75).46 The patient did not fulfil the criteria for brainstem death, although there was some impairment of brainstem function clinically. On initial assessment, halo traction or any other surgery was not appropriate in view of the complete nature of the spinal cord injury, although he had obvious instability at the occipito/cervical level. Early stabilization, which is the recommended management for AOD,42 would have no effect on clinical outcome. Moreover, any kind of surgery and attempts of reduction would be an additional life-threatening risk. The position of the occipito–cervical junction has not been changed, owing to the risk of vascular damage and resultant catastrophic cerebral haemorrhage.47 The prognosis for any recovery was virtually nonexistent; at best, he would require continuous life support and ventilation.
Neurological examination revealed that this patient had a syndrome similar to LIS.14 LIS is caused by a primary vascular or traumatic injury to the brainstem, and characterized by upper motor neuron quadriplegia, paralysis of lower cranial nerves, bilateral paresis of horizontal gaze and anarthria. It is typically associated with a ventral pons lesion owing to obstruction of the basilar artery. Patients with such pontine lesions are conscious; they generally have preserved vertical eye movements and movement of the eyelids (blinking), this being their only means of responding to the outside world.48 It is somewhat difficult to immediately differentiate LIS from other types of upper nervous system damage. Although our patient with a complete medulla cervical spinal cord transection had nil likelihood of functional recovery, there have been reports of patients who present with LIS or similar dense neurologic deficit at the craniocervical level with preservation of structure that have surprising degrees of recovery.49, 50 It is commonly assumed that the quality of life of a patient with LIS is so poor that it is not worth living. However, the contrary is exposed in the work by Jean-Dominique Bauby, ‘The Diving Bell and the Butterly: A Memoir of Life in Death’. In this remarkable book, the author who is afflicted with LIS laboriously dictated his experience by blinking.
Since his injury, our patient has spent 101 days in ICU, being 50 at the initial admitting hospital and a further 51 days within the trauma center at Royal Perth Hospital. Thereafter, he was transferred to the Royal Perth Hospital rehabilitation campus. It is believed that this patient will eventually be discharged from hospital and into the community, albeit with necessary life-sustaining treatment, including medical observation, artificially delivered fluids, nutrition and ventilator support. Nevertheless, a medical decision needs to be made regarding the short- and long-term management of this 21-year-old male who has no prospect of breathing spontaneously or gaining any upper or lower limb function. It will need to take into account legal and ethical issues, and be made in accord with the patient and his immediate family's needs and wishes.
Cases such as this raise significant ethical questions. For example, is our patient in such a devastating state able to judge his situation appropriately, and who will decide whether he is competent. For incompetent patients, it is of paramount importance that medical specialists fight for their interests. Along with accepting the right of the patient to die comes the uncertainty as to whether or not the patient might change his position at a later time. In patients with permanent brain damage, life and death decisions clearly fall to physicians, family members, guardians or other surrogates for the patient. In our case, in agreement with his family, we deemed our patient to be competent to make decisions about his ongoing life support. He was asked about whether he wanted to be kept alive and he indicated in the affirmative, several times, by his way of communicating by eye blinking. Recently, however, he developed severe depression and is now receiving antidepressant medication. In light of the deterioration of his mental health, the question arises as to where he is likely to change his mind about remaining of life support and, in turn, whether he is now competent to make such a decision.
Conclusion
This is the first known reported patient to survive AOD with a complete transection of the medulla/cervical spine level, theoretically, an unsurvivable injury. The consequences are formidable. In addition to the emotional and financial impact on the family, this injury is associated with long-term hospitalization, including prolonged ICU stay and severe life-threatening complications. This equates to high social and economic costs in a patient without any prospect of functional recovery. Further consideration will be required in regard to expected quality of life in such surviving patients. In cases like this, the emotional, financial and medical consequences for the patient, family and medical staff would require close scrutinization of the allocation of limited resources.
References
Imaizumi T, Sohma T, Hotta H, Teto I, Imaizumi H, Kaneko M . Associated injuries and mechanism of atlanto-occipital dislocation caused by trauma. Neurol Med Chir (Tokyo) 1995; 35: 385–391.
Adams VI . Neck injuries: I. Occipitoatlantal dislocation – a pathologic study of twelve traffic fatalities. J Forensic Sci 1992; 37: 556–564.
Deliganis AV et al. Radiologic spectrum of craniocervical distraction injuries. Radiographics 2000; 20: S237–S250.
Gregg S, Kortbeek JB, du Plessis S . Atlanto-occipital dislocation: a case study of survival with partial recovery and review of the literature. J Trauma 2005; 58: 168–171.
Palmer MT, Turney SZ . Tracheal rupture and atlanto-occipital dislocation: case report. J Trauma 1994; 37: 314–317.
Payer M, Sottas CC . Traumatic atlanto-occipital dislocation: presentation of a new posterior occipitoatlantoaxial fixation technique in an adult survivor: technical case report. Neurosurgery 2005; 56: E203.
Lee C, Woodring JH, Walsh JW . Carotid and vertebral artery injury in survivors of atlanto-occipital dislocation: case reports and literature review. J Trauma 1991; 31: 401–407.
Kaufman RA, Dunbar JS, Botsford JA, McLaurin RL . Traumatic longitudinal atlanto-occipital distraction injuries in children. AJNR Am J Neuroradiol 1982; 3: 415–419.
Rao G, Arthur AS, Apfelbaum RI . Circumferential fracture of the skull base causing craniocervical dislocation. Case report. J Neurosurg 2002; 97: 118–122.
Bools JC, Rose BS . Traumatic atlanto-occipital dislocation: two cases with survival. AJNR Am J Neuroradiol 1986; 7: 901–904.
Steinmetz MP, Lechner RM, Anderson JS . Atlanto-occipital dislocation in children: presentation, diagnosis, and management. Neurosurg Focus 2003; 14: ecp1.
Werne S . Studies in spontaneous atlas dislocation. Acta Orthop Scand 1957; (Suppl 23): 1–150.
Grob D . Transarticular screw fixation for atlanto-occipital dislocation. Spine 2001; 26: 703–707.
Woodring JH, Selke AC, Duff DE . Traumatic atlanto-occipital dislocation with survival. AJR Am J Roentgenol 1981; 137: 21–24.
Sponseller PD, Cass JR . Atlanto-occipital fusion for dislocation in children with neurologic preservation. A case report. Spine 1997; 22: 344–347.
Guigui P, Milaire M, Morvan G, Lassale B, Deburge A . Traumatic atlanto-occipital dislocation with survival: case report and review of the literature. Eur Spine J 1995; 4: 242–247.
Harmanli O, Koyfman Y . Traumatic atlanto-occipital dislocation with survival: a case report and review of the literature. Surg Neurol 1993; 39: 324–330.
Hadley MN, Dickman CA, Browner CM, Sonntag VK . Acute traumatic atlas fractures: management and long term outcome. Neurosurgery 1988; 23: 31–35.
Ahuja A, Glasauer FE, Alker GJ, Klein DM . Radiology in survivors of traumatic atlanto-occipital dislocation. Surg Neurol 1994; 41: 112–118.
Papadopoulos SM, Dickman CA, Sonntag VK, Rekate HL, Spetzler RF . Traumatic atlanto-occipital dislocation with survival. Neurosurgery 1991; 28: 574–579.
Fruin AH, Pirotte TP . Traumatic atlanto-occipital dislocation. Case report. J Neurosurg 1977; 46: 663–666.
Bohlman HH . Acute fractures and dislocations of the cervical spine. An analysis of three hundred hospitalized patients and review of the literature. J Bone Joint Surg Am 1979; 61: 1119–1142.
Hadley MN . Diagnosis and management of traumatic atlanto-occipital dislocation injuries. [guidelines for the management of acute cervical spine and spinal cord injuries: Chapter 14]. Neurosurgery 2002; 50: S105–S113.
Traynelis VC, Marano GD, Dunker RO, Kaufman HH . Traumatic atlanto-occipital dislocation. Case report. J Neurosurg 1986; 65: 863–870.
Belzberg AJ, Tranmer BI . Stabilization of traumatic atlanto-occipital dislocation. Case report. J Neurosurg 1991; 75: 478–482.
Dickman CA, Papadopoulos SM, Sonntag VK, Spetzler RF, Rekate HL, Drabier J . Traumatic occipitoatlantal dislocations. J Spinal Disord 1993; 6: 300–313.
Baker SP, O'Neill B, Haddon W, Long WB . The injury severity score: a method for describing patients with multiple injuries and evaluating emergency care. J Trauma 1974; 14: 187–196.
Chattar-Cora D, Valenziano CP . Atlanto-occipital dislocation: a report of three patients and a review. J Orthop Trauma 2000; 14: 370–375.
Henry MB, Angelastro DB, Gillen JP . Unrecognized traumatic atlanto-occipital dislocation. Am J Emerg Med 1998; 16: 406–408.
Alker Jr GJ, Oh YS, Leslie EV . High cervical spine and craniocervical junction injuries in fatal traffic accidents: a radiological study. Orthop Clin North Am 1978; 9: 1003–1010.
Powers B, Miller MD, Kramer RS, Martinez S, Gehweiler JA . Traumatic anterior atlanto-occipital dislocation. Neurosurgery 1979; 4: 12–17.
Dublin AB, Marks WM, Weinstock D, Newton TH . Traumatic dislocation of the atlanto-occipital articulation (AOA) with short-term survival. With a radiographic method of measuring the AOA. J Neurosurg 1980; 52: 541–546.
Junge A, Krueger A, Petermann J, Gotzen L . Posterior atlanto-occipital dislocation and concomitant discoligamentous C3-C4 instability with survival. Spine 2001; 26: 1722–1725.
Bucholz RW, Burkhead WZ . The pathological anatomy of fatal atlanto-occipital dislocations. J Bone Joint Surg Am 1979; 61: 248–250.
Chaljub G, Singh H, Gunito FC, Crow WN . Traumatic atlanto-occipital dislocation: MRI and CT. Neuroradiology 2001; 43: 41–44.
Pang D, Wilberger JE . Traumatic atlanto-occipital dislocation with survival: case report and review. Neurosurgery 1980; 7: 503–508.
Piatt JH . Trauma case for diagnosis: atlanto-occipital dislocation. Mil Med 1989; 154: 455–457.
Fujimura Y, Nishi Y, Chiba K, Kobayashi K . Prognosis of neurological deficits associated with upper cervical spine injuries. Paraplegia 1995; 33: 195–202.
Saeheng S, Phuenpathom N . Traumatic occipitoatlantal dislocation. Surg Neurol 2001; 55: 35–40.
Labler L, Eid K, Platz A, Trentz O, Kossmann T . Atlanto-occipital dislocation: four case reports of survival in adults and review of the literature. Eur Spine J 2004; 13: 172–180.
Champion HR, Sacco WJ, Carnazzo AJ, Copes W, Fouty WJ . Trauma score. Crit Care Med 1981; 9: 672–676.
Przybylski GJ, Clyde BL, Fitz CR . Craniocervical junction subarachnoid hemorrhage associated with atlanto-occipital dislocation. Spine 1996; 21: 1761–1768.
Kenter K, Worley G, Griffin T, Fitch RD . Pediatric traumatic atlanto-occipital dislocation: five cases and a review. J Pediatr Orthop 2001; 21: 585–589.
Fisher CG, Sun JC, Dvorak M . Recognition and management of atlanto-occipital dislocation: improving survival from an often fatal condition. Can J Surg 2001; 44: 412–420.
Seibert PS et al. Cranio-cervical stabilization of traumatic atlanto-occipital dislocation with minimal resultant neurological deficit. Acta Neurochir (Wiener) 2005; 147: 435–442.
Ferrera PC, Bartfield JM . Traumatic atlanto-occipital dislocation: a potentially survivable injury. Am J Emerg Med 1996; 14: 291–296.
Feiz-Erfan I, Gonzalez LF, Dickman CA . Atlanto-occipital transarticular screw fixation for the treatment of traumatic occipitoatlantal dislocation. Technical note. J Neurosurg Spine 2005; 2: 381–385.
Laureys S et al. The locked-in syndrome: what is it like to be conscious but paralysed and voiceless? Prog Brain Res 2005; 150: 495–511.
McCusker EA, Rudick RA, Honch GW, Griggs RC . Recovery from the ‘locked-in’ syndrome. Arch Neurol 1982; 39: 145–147.
Silver B, Grover KM, Arcila X, Mitsias PD, Bowyer SM, Chopp M . Recovery in a patient with locked-in syndrome. Can J Neurol Sci 2006; 33: 246–249.
Author information
Authors and Affiliations
Additional information
No sources of financial support
Rights and permissions
About this article
Cite this article
Gautschi, O., Woodland, P. & Zellweger, R. Complete medulla/cervical spinal cord transection after atlanto-occipital dislocation: An extraordinary case. Spinal Cord 45, 387–393 (2007). https://doi.org/10.1038/sj.sc.3101975
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.sc.3101975