Abstract
Study design:
Retrospective case series.
Objectives:
To analyze prognostic factors for patients with spinal cord astrocytomas.
Setting:
Department of Orthopaedic Surgery, Keio University, Japan.
Methods:
Seven patients received total excisions (group T), eight underwent partial excisions (group P), and 15 had excisional biopsies (group B). Impacts of the tumor histological grade, the level of the tumor, the types of surgical interventions, and the use of adjuvant radiotherapies on the survival and functional outcomes of 30 patients (18 in low-grade and 12 high-grade malignancy tumors) were analyzed.
Results:
The survival rate of the low-grade malignancy group was significantly higher than that of the high-grade group. The survival rate of the patients with thoracic astrocytomas was significantly higher than those with cervical astrocytomas. In both the low- and high-grade groups, the survival rates in groups P/T were significantly higher than those in group B. In the low-grade group, five patients, whose preoperative functional statuses were classified as ‘fair’ or better, remained ‘fair’ or better after surgery. In the high-grade group, the postoperative functional statuses were classified as ‘no change’ or ‘aggravated’ in all except two patients. No significant difference in the survival rates was detected between patients with and without adjuvant radiotherapy.
Conclusions:
The tumor grade and the extent of tumor resection were significant prognostic factors for survival rate. In low-grade malignancy group, good motor function was retained when surgeries were performed before substantial neurological deterioration. The efficacy of postoperative radiotherapy has yet to be determined and needs further study.
Similar content being viewed by others
Introduction
Intramedullary spinal cord astrocytomas represent only 6–8% of spinal cord tumors.1, 2, 3 Because of the rarity of this tumor, optimal treatment guidelines have yet to be defined. The traditional treatment strategy for intramedullary astrocytoma is a biopsy to confirm the diagnosis followed by radiation therapy.4 However, the efficacy of this treatment has been a subject of controversy. Important but presently unsettled issues include the impact of the extent of resection on local control and survival1, 5, 6 and the significance of postoperative radiation therapy.7 The purpose of the present study was to obtain useful information to establish decision-making guidelines for the treatment of spinal cord astrocytoma by examining the clinical records of patients treated at Keio University Hospital.
Materials and methods
The clinical outcomes of 30 patients who underwent surgical treatments for astrocytomas at Keio University Hospital between 1986 and 2002 were reviewed. Among these 30 patients, 17 subjects were male and 13 were female; the patients’ age ranged from 7 to 50 years at the time of surgery, with a mean of 35 years. The follow-up period ranged from 1 to 16.5 years, with a mean of 5.5 years. The pathological grades of the tumors were decided according to Kernohan's classification system.8 Grade I tumors were present in seven patients, grade II tumors in 11 patients, grade III tumors in 11 patients, and grade IV tumors in one patient. The tumors were located at the cervical level in 13 patients, at the thoracic level in 16 patients, and at the conus medullaris in one patient. Among the 30 patients enrolled in the present study, an excisional biopsy was performed in 15 patients (group B), a partial resection in eight patients (group P), and a total resection in seven patients (group T). Adjuvant radiotherapy was administered to 19 patients in whom a macroscopic total resection could not be accomplished. The median dosage was 45 Gy (range, 40–55 Gy) in 25 fractions (range, 20–34 fractions) delivered over 4–6 weeks. The median dose per fraction was 1.8 Gy (range, 1.5–2.0 Gy). In the present study, the impacts of the tumor grade, the level of the tumor, the type of surgical intervention, and the use of adjuvant radiotherapy on the survival and functional outcomes of the patients were analyzed.
The patients were divided into two groups based on the pathological grade of the tumor: a low-grade malignancy group (grades I and II), and a high-grade malignancy group (grades III and IV). Survival was the period from initial surgery to the date of death or last follow-up. Survival curves were drawn using the Kaplan–Meier method.9 Potentially significant survival differences were assessed utilizing the Wilcoxon test.10 The functional status of each patient was determined using the method described by Guidetti et al,11 which is a four-stage system: excellent (E), good (G), fair (F), and poor (P).
Results
Overview of surgical outcomes
Among the 30 patients, 21 subjects had died before the start of the present study (Table 1). The survival rate for all 30 patients with spinal cord astrocytoma was 68% at 5 years and 36% at 10 years (Figure 1a). The causes of death were central respiratory failure due to tumor progression in 12 patients and chronic infection (pneumonia, urinary tract infection) in five patients; the onset of these conditions was greatly facilitated by development or progression of paralysis. In the remaining four patients who died before the start of the study, two subjects died of coma and two subjects died of unknown causes. Among the nine survivors, seven patients had low-grade malignancies and the remaining two patients had high-grade malignancies. One of the two surviving patients in the high-grade malignancy group responded completely to radiotherapy after a biopsy.
Predictors of survival
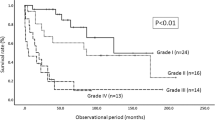
The survival rate of patients in the low-grade malignancy group was significantly higher than that in the high-grade malignancy group (P=0.0011) (Figure 1b). The survival rate of the patients with thoracic astrocytomas was significantly higher than that of the patients with cervical astrocytomas in both the low-grade (P=0.0251) and high-grade (P=0.0126) malignancy groups (Figure 2a and b). With regard to the type of surgical intervention, in both the low-grade and high-grade malignancy groups, the survival rate of group P/T was significantly higher than that of group B (low-grade: P=0.0251, high-grade: P=0.0342) (Figure 3a and b).
Radiotherapy was administered to patients in whom a macroscopic total resection could not be accomplished; however, this treatment was abandoned in five patients (four in group B and one in group P) because of postoperative progression of paralysis or deterioration in the patient's general condition (Table 2). With regard to the survival rate, difference between radiation and non-radiation groups was not significant in either the low-grade or the high-grade malignancy group (Figure 4a and b).
Correlation between type of surgical procedure and functional outcome
We analyzed the effect of the type of surgical intervention on functional outcome. The preoperative functional statuses of the patients in the low-grade malignancy group were excellent in seven, good in one, fair in five, and poor in five patients. Only five patients (one from group B and two each from groups P and T) were judged as ‘fair’ or better after surgery. In these five patients, the preoperative functional statuses were ‘fair’ or better (Figure 5). In the high-grade malignancy group, the preoperative functional statuses were excellent in two patients, good in one patient, fair in six patients, and poor in three patients. The postoperative functional statuses were classified as ‘no change’ or ‘aggravated’ in all of the patients in this group except for two patients. One patient responded completely to radiotherapy after a biopsy, whereas the second patient experienced a temporary neurological deterioration after a total resection who subsequently exhibited a good functional recovery when examined at a 2-year follow-up. Except for these two patients, all the patients in the high-grade malignancy group died before the completion of this study.
Case 1
A 32-year-old male had developed numbness in his upper limbs 2 years before surgery. A preoperative neurological assessment revealed a motor disturbance in his left upper extremity. Preoperative T1- and T2-weighted magnetic resonance (MR) images revealed diffuse swelling of the spinal cord with iso- and high-signal intensities with poor delineation in the upper cervical region (Figure 6a and b), and gadolinium (Gd)-enhanced T1-weighted MR images demonstrated a partial signal enhancement (Figure 6c). As the tumor was not well delineated and an intraoperative biopsy established a diagnosis of a grade III astrocytoma, further surgical intervention was abandoned; the patient was treated with postoperative radiotherapy. The lesion gradually invaded the medulla oblongata (Figure 6d–f), and the patient died from central respiratory failure 15 months after the biopsy.
Case 2
A 23-year-old male developed numbness in his lower extremities and a slight gait disturbance 6 months before surgery. Preoperative T1- and T2-weighted MR images revealed extensive swelling of the spinal cord with iso- and high-signal intensities at the cervico-thoracic level (Figure 7a and b). As the tumor was delineated and an intraoperative biopsy established a diagnosis of a grade II astrocytoma, total resection was performed. At 5 years after the surgery, there was no sign of tumor recurrence on MR images (Figure 7c and d) and he returned to the normal life with no clinical symptom in spite of the severe spinal cord atrophy.
Discussion
Predictors for better survival
As documented in previous studies,2, 3, 12 the most significant predictor of survival in patients with spinal cord astrocytoma is the pathological grade of the tumor. In the present study, patients who had tumors located in the thoracic cord had a significantly better prognosis than those with tumors located in the cervical cord. This finding can probably be explained by the assumption that it would take a thoracic tumor longer to reach the upper cervical cord, eventually causing central respiratory failure, the leading cause of death in patients with spinal astrocytomas.
No consensus has been made as to the most appropriate surgical intervention for the treatment of spinal cord astrocytomas. Some investigators recommend aggressive resection of the tumor,1, 12 whereas others suggest that biopsy alone yielded a better prognosis.2, 3, 13 However, most of the studies that do not recommend tumor resection were conducted before the introduction of technological innovations such as magnetic resonance imaging, the Cavitron Ultrasonic Surgical Aspirator (CUSA), and intraoperative spinal cord monitoring. With these technological advances in mind, the clinical debate regarding appropriate treatment for spinal cord astrocytomas should be resumed.14
In the present study, total and partial resections yielded better survival rates than biopsy alone in patients with spinal astrocytomas. Furthermore, a higher survival rate can be expected for cases with thoracic astrocytomas, compared to those with cervical astrocytomas. However, we have to analyze these results carefully because this is a retrospective study, even though it would be impossible to conduct a prospective study. A bias in indications of surgical interventions cannot be ruled out. Although we always attempted to perform total resection initially, it is not always possible to accomplish the procedure owing to the size, location, and nature of tumors. Because recent reports demonstrated that pilocytic astrocytomas can be totally resected, even if they look like high-grade malignancy tumors,14 we should attempt at least partial, if possible total, resection of the tumor, especially for thoracic astrocytomas.
Predictors for better functions
Pre- and postoperative evaluations of the patients’ motor performances revealed that (1) five out of nine patients (four with low-grade malignancy tumors and one with a high-grade malignancy tumor) whose functional status was rated as ‘fair’ or better before surgery remained at ‘fair’ or better functional status after partial or total resection, (2) no improvements were observed in patients whose preoperative functional status was judged to be poor, regardless of the type of surgical procedure performed, and (3) four patients whose preoperative functional statuses were judged to be excellent and who underwent a biopsy alone had a poor functional outcome or died during the period of the study. Based on these findings, partial or total tumor resection should be considered in patients with low-grade astrocytomas, as a favorable postoperative functional outcome can be expected if the preoperative motor function of the patient is still preserved.15
Selection of a biopsy alone to avoid neurological deterioration may not be appropriate even for patients with high-grade malignancy astrocytomas, as both the functional outcomes and the survival rates of patients were poor after this procedure. On the other hand, partial and total resections of the tumor yielded a better survival rate, although the number of patients who underwent a total resection was relatively small. Taken together, the aforementioned prognostic suggests that partial or total resection should be attempted, even if these approaches may sometimes lead to a poor functional outcome immediately after surgery. A cordotomy might be justified in some cases of highly malignant tumors if the tumor is located at the thoracic level to obtain better survival rate.
Effect of radiotherapy
The efficacy of radiotherapy for the treatment of spinal cord astrocytomas has yet to be determined.3, 15, 16, 17 In the present study, no significant difference in survival rates was observed between patients who received adjuvant radiotherapy and those who did not, regardless of the pathological grades. Radiotherapy was administered to patients with residual tumors after surgical resection but was abandoned in five patients because of the progression of postoperative paralysis or deterioration in the patient's general condition. As such bias in the indications for radiotherapy may have confounded the results, the efficacy of radiotherapy for the treatment of spinal cord astrocytomas could not be determined in the present study. Interestingly, however, one patient with a high-grade astrocytoma responded completely to radiotherapy. A prospective study to clarify the effectiveness of radiotherapy for the treatment of spinal cord astrocytomas is presently being planned.
Conclusion
The histological grade of the tumor was the most significant prognostic factor of survival. The level of tumor and the extent of tumor resection were significantly correlated with patients’ survival. Good motor function was retained when surgeries were performed before substantial neurological deterioration in low-grade malignancy astrocytomas. The efficacy of postoperative radiotherapy has yet to be determined and needs further study.
References
Epstein FJ, Farmer JP, Freed D . Adult intramedullary astrocytomas of the spinal cord. J Neurosurg 1992; 77: 355–359.
Innocenzi G, Salvati M, Cervoni L, Delfini R, Cantore G . Prognostic factors in intramedullary astrocytomas. Clin Neurol Neurosurg 1997; 99: 1–5.
Minehan KJ, Shaw EG, Scheuthauer BW, Davis DL, Onofrio BM . Spinal cord astrocytoma: pathological and treatment considerations. J Neurosurg 1995; 83: 590–595.
Marsa GW, Goffine DR, Rubinstein LJ, Bagshaw MA . Megavoltage irradiation in the treatment of gliomas of the brain and spinal cord. Cancer 1975; 36: 1681–1689.
Cooper PR . Outcome after operative treatment of intramedullary spinal cord tumors in adults: intermediate and long-term results in 51 patients. Neurosurgery 1989; 25: 855–859.
Epstein F, Epstein N . Surgical treatment of spinal cord astrocytoma of childhood. A series of 19 patients. J Neurosurg 1982; 57: 685–689.
Kopelson G, Linggood RM, Kleinman GM, Doucette J, Wang CC . Management of intramedullary spinal cord tumors. Radiology 1980; 135: 473–479.
Kernohan JW, Mabon RF, Svein HJ . Symposium on new and simplified concept of gliomas: simplified classification of the gliomas. Mayo Clin Proc 1949; 24: 71–75.
Kaplan EL, Meier P . Nonparametric estimation from incomplete observations. J Am Stat Assoc 1958; 53: 457–481.
Mantel N . Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep 1966; 50: 163–170.
Guidetti B, Mercuri S, Vagnozzi R . Long-term results of the surgical treatment of 129 intramedullary spinal gliomas. J Neurosurg 1981; 54: 323–330.
Cooper PR, Epstein F . Radical resection of intramedullary spinal cord tumors in adults. Recent experience in 29 patients. J Neurosurg 1985; 63: 492–499.
Sandler HM, Papadopoulis SM, Thornton AF, Ross DA . Spinal cord astrocytomas: results of therapy. Neurosurgery 1992; 30: 490–493.
Brotchi J et al. A survey of 65 tumors within the spinal cord: surgical results and the importance of preoperative magnetic resonance imaging. Neurosurgery 1991; 29: 651–657.
Kim MS, Chung CK, Choe G, Kim IH, Kim HJ . Intramedullary spinal cord astrocytoma in adults: postoperative outcome. J Neurooncol 2001; 52: 85–94.
Jyothirmayi R, Madhavan J, Nair MK, Rajan B . Conservative surgery and radiotherapy in the treatment of spinal cord astrocytoma. J Neurooncol 1997; 33: 205–211.
Rodrigues GB, Waldron JN, Wong CS, Laperriere NJ . A retrospective analysis of 52 cases of spinal cord glioma managed with radiation therapy. Int J Radiate Oncol Biol Phys 2000; 48: 837–842.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nakamura, M., Chiba, K., Ishii, K. et al. Surgical outcomes of spinal cord astrocytomas. Spinal Cord 44, 740–745 (2006). https://doi.org/10.1038/sj.sc.3101932
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.sc.3101932
Keywords
This article is cited by
-
Clinical outcomes and a therapeutic indication of intramedullary spinal cord astrocytoma
Spinal Cord (2022)
-
Predictors of survival and time to progression following operative management of intramedullary spinal cord astrocytomas
Journal of Neuro-Oncology (2022)
-
Adult primary high-grade spinal glioma: a nationwide analysis of current trends in treatment and outcomes
Journal of Neuro-Oncology (2020)
-
Predictors of mortality in patients with primary spinal cord glioblastoma
European Spine Journal (2020)
-
Impact of surgery and radiation therapy on spinal high-grade gliomas: a population-based study
Journal of Neuro-Oncology (2018)