Abstract
Objective: Documentation of MRI and neurophysiological changes following accidental electrical injury.
Setting: Tertiary care referral teaching hospital at Lucknow, India.
Results: A 30-year-old lady developed amnesia and spastic paraparesis with loss of pin prick sensation below the second thoracic spinal segment following electrocution. Her spinal MRI was normal and cranial MRI revealed T2 hyperintensity in the right putamen. Peroneal, sural and electromyography were normal. Tibial central sensory conduction time was normal but central motor conduction time to lower limbs and right upper limb was prolonged.
Conclusion: Neurophysiological study and MRI may help in understanding the pathophysiological basis of neurological sequelae following electrical injury.
Similar content being viewed by others
Introduction
Rural electrification has received great attention from the government for improving agricultural and small scale industry development in India. This has inherent hazards because of the ignorance of villagers and poor maintenance of electrical cables. This results in several electrical accidents caused by the touching of live wires. Often such accidents go unreported. Electrical injury may result in various acute and delayed neurological complications such as unconsciousness, amnesia, seizure, choreoathetosis, cerebellar ataxia, parkinsonism, myelopathy and peripheral neuropathy.1 Various structural and functional abnormalities following electrocution can be studied by the help of MRI and neurophysiological evaluation. There is a paucity of reports correlating clinical, MRI and neurophysiological changes in electrocution.1,2,3 We recently managed a patient with electrical injury caused by touching a high-tension wire lying in her flooded field. We report the clinical, MRI and electrophysiological findings of this patient and highlight the sequelae of neurological complications following electrocution.
Case Report
A 30-year-old lady had an electrical injury in June 1999, when she was lifting an electrical wire lying across the road. Her hands were wet, the road was flooded with water and the wire was conducting AC of 11 000 V. Immediately, she had fallen down and the wire stuck to her chest. The current flow was discontinued after about 5 min and she was discovered in an unconscious state. Her left thumb and index finger were severely charred. She was taken to the local hospital where she was found to have sustained a 35% burn localised to neck, chest, both hands and left foot. Once she regained consciousness after 24 h, she was found to have amnesia about the event and weakness and loss of sensation of lower limbs. She did not have a seizure and there were no symptoms related to bowel and bladder dysfunction. Gradually her lower limb power improved and she was able to sit independently by 2 months and walk with support by 4 months. She also started perceiving sensations in the lower limbs. Her right thumb, index and middle fingers were amputated at 1 month. She was brought to our hospital 13 months after electrocution because of residual neurological deficit.
On examination, she had scar marks on neck, chest, hands and left foot. The right thumb and index finger and left thumb, index and middle fingers had been amputated. Her pulse was 84/min, regular and BP 120/80 mmHg. She had mild pallor. Cardiovascular, respiratory and abdominal examinations were normal. She had total amnesia about the electrocution. She was oriented to time, place and person. She had reduced attention span and impairment of recent and immediate memory. Her remote memory was normal. Other mental faculties were normal and consistent with her socio-economic and educational background. There was no cranial nerve palsy and no Horner's sign. She had a spastic paraparesis of grade 4. Biceps, triceps and knee reflexes were brisk and there was ankle clonus bilaterally. Plantar responses were extensor on both sides. Pin prick and touch sensations were reduced below the second thoracic spinal level. Sense of position and movement were normal and there was no cerebellar sign.
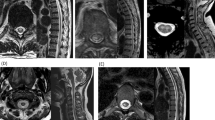
Investigations revealed normal haemogram, blood counts and serum chemistry. Electrocardiogram was normal. Radiograph of chest and cervical spine was normal. Craniospinal MRI was done in a 2T scanner operating at 1.5 T and revealed small hyperintense signal changes on T2 on the right putamen (Figure 1a). Spinal MRI was normal. Nerve conduction studies of sural, peroneal, median and ulnar motor and sensory were normal. F-response latencies of upper and lower limbs were normal. Concentric needle electromyography of abductor pollicis brevis, extensor digitorum communis, biceps and tibialis anterior was normal. Median and tibial somatosensory evoked potentials were carried out by stimulating the median nerve at the wrist and tibial nerve at the ankle and recording from the contralateral parietal cortex and Erb's point for median SEP and vertex and first lumbar spine for tibial. The difference of cortical and spinal latency was used to calculate the central sensory conduction time. Central motor conduction was carried out by stimulating the respective motor area and recording from abductor digiti minimi for the upper limb and tibialis anterior for the lower limb in a belly tendon montage. The spinal stimulation was applied at the seventh cervical and first lumbar vertebra respectively. Central motor conduction was calculated by subtracting the spinal latency from that of cortical. The values were compared with our laboratory normal data.4 Central sensory conduction time of median (right=8.4 ms; left=8.6 ms) and tibial (right=25 ms; left=26.0 ms) were within normal limits. Central motor conduction time to right abductor digiti minimi was prolonged (10.4 ms) whereas to left it was normal (7.2 ms). Central motor conduction time to tibialis anterior was markedly prolonged being 38.0 ms on left and 25.2 ms on right side (Figure 1b). She was advised physiotherapy and baclofen 5 mg thrice daily for spasticity.
Discussion
Our patient with severe electrocution developed spastic paraparesis and memory impairment. The factors that determine the anatomic pattern, the extent of tissue injury and relative contribution of heat versus direct electrical damage include the magnitude of onset, frequency of current, anatomic location and contact duration. With more prolonged contact in the range of seconds, thermal damage in the subcutaneous tissue becomes substantial. Vulnerability of different tissues to supraphysiological temperature is similar. Our patient also received electrical injury while holding a high tension wire but she was in a flooded road which may be responsible for relatively lesser degree of burns in her hands and still lesser in her feet. Low resistance because of wet hands and feet could have resulted in a milder degree of burn and also sparing of posterior columns. Sparing of proprioceptive pathways in our patient was evidenced by normal joint position and vibration sensations and somatosensory evoked potential. In an earlier study both median and tibial SEPs were reported to be prolonged following electrocution.2 It is possible that their patient might have suffered from more severe electrocution which necessitated below knee amputation of both legs. Central motor conduction time was prolonged markedly to the lower limbs and moderately to the right upper limb. This may be consistent with demyelination of motor pathways. However desynchronisation of descending volleys may also result in mild prolongation of central motor conduction time. In autopsy study, following lightning injury, demyelination of spinal cord has been reported.5 Impaired pinprick and touch below the second thoracic spinal level in our patient suggests spinal cord involvement; however, spinal MRI was normal. This may be due to low sensitivity of MRI in detecting spinal cord demyelination on one hand and delay of 13 months in carrying out MRI on the other. Cranial MRI of our patient however revealed small discrete hyperintense signal changes on T2 in the right putamen. In another study after 4–8 weeks of electrical injury, MRI was reported to be normal.3 In three patients with lightning injury, hyperintense signal changes in the basal ganglia were found in one and cerebellar atrophy in the other and in the third patient MRI was normal.6
In the acute stage, the spinal cord changes include haematoma, cord oedema and petechial haemorrhages. In the brain, there are multifocal petechial haemorrhages throughout the cerebral cortex and medulla with widespread chromatolysis.7 In our study, EMG was not suggestive of anterior horn cell involvement. Current exceeding 50 mA which passes across the hands or hand to feet may result in cardiac arrhythmia especially if current flows during early myocardial repolarisation. Unconsciousness in victims of electrical injury may be due to cardiac arrhythmia or respiratory arrest or metabolic alterations. Our patient was unconscious for 24 h and even now amnesic about the event. The presence of hyperintense lesions in the putamen are consistent with cerebral anoxia which may be attributed to respiratory arrest or cardiac arrhythmia or both. Memory loss may be due to hippocampal involvement which is most vulnerable to anoxic injury.
The electrical injury may commonly result in neurological morbidity especially in the developing countries where high voltage currents are used and the infrastructure for maintenance of transmitting cables are inadequate.
References
Howkes CH, Thorpe JW . Acute polyneuropathy due to lightning injury J Neurol Neurosurg Psychiat 1992 55: 338–390
Triggs WJ et al. Central conduction abnormalities after electrical injury Muscle Nerve 1994 17: 1068–1070
William J et al. Central conduction abnormalities after electrical injury Muscle Nerve 1994 17: 1068–1070
Kalita J, Misra UK . Neurophysiological studies in acute transverse myelitis J Neurol 2000 247: 943–948
Davidson GS, Deck JH . Delayed myelopathy following lightning strike: a demyelinating process Acta Neuropathol 1988 77: 104–108
Cherington M, Yarnell P, Hallmark D . MRI in lightning encephalopathy Neurology 1993 43: 1437–1438
Crichley M . Neurological effects of lightning and electricity Lancet 1934 1: 68072
Acknowledgements
We thank Mr Rakesh Kumar Nigam for preparation of manuscript.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kalita, J., Jose, M. & Misra, U. Myelopathy and amnesia following accidental electrical injury. Spinal Cord 40, 253–255 (2002). https://doi.org/10.1038/sj.sc.3101275
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.sc.3101275
Keywords
This article is cited by
-
Cervicothoracic spinal cord and pontomedullary injury secondary to high-voltage electrocution: a case report
Journal of Medical Case Reports (2012)