Abstract
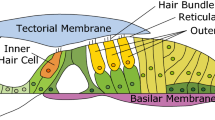
The primary sensory hair cells of the mammalian cochlea are located in the organ of Corti, a sensory epithelium which separates fluids of widely differing chemical composition. The apical, sensory surfaces of the hair cells are exposed to the potassium-rich endolymph of the scala media and their lateral and ventral surfaces are exposed to the perilymph of the scala tympani whose chemical composition resembles that of other extracellular fluids1–3. The high potassium concentration of the endolymph (150 mM) is believed to result from the activity of electrogenic potassium pumps located in the stria vascularis which lines the lateral walls of the cochlea3. These pumps are also the source of the positive endocochlear potential of about +80 mV which can be recorded from the scala media4–6. When the ear is stimulated with sound, receptor potentials may be recorded extracellularly from the fluid-filled spaces of the cochlea7,8, and intracellularly from the rows of inner and outer hair cells9–11. According to the ‘resistance microphone’ theory of Davis12, these receptor potentials are derived from the preexisting polarization of the hair cells by a change in the ohmic resistance of the mechanosensitive portion of the cell membrane (Fig. 1). This produces potential changes in the scala tympani and scala media of opposite phase, thus giving rise to the cochlear microphonic (CM). Evidence is presented here to support this theory. When sufficient depolarizing current is injected into inner hair cells to cancel the polarizing voltage, the receptor potentials disappear, and their phase is reversed when the polarizing voltage across the apical membranes of the hair cells is reversed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Smith, C. A., Lowry, O. H. & Wu, M. L. Laryngoscope 64, 141–153 (1954).
Bosher, S. K. & Warren, R. L. Proc. R. Soc. B171, 227–247 (1968).
Johnston, B.M. & Sellick, P. M. Q. Rev. Biophys. 5, 1–57 (1972).
Tasaki, I. & Spyropoulos, C. S. J. Neurophysiol. 22, 149–155 (1959).
Kuipers, W. & Bonting, S. L. Biochim. biophys. Acta 173, 477–485 (1969).
Kuipers, W. & Bonting, S. L. Plügers Arch. ges. Physiol. 320, 348–358 (1970).
Wever, E. G. & Bray, C. W. Proc. Acad. natn. Sci. U.S.A. 16, 344–350 (1930).
Dallos, P. The Auditory Periphery (Academic, New York, 1973).
Russell, I. J. & Sellick, P. M. Nature 267, 858–860 (1977).
Tanaka, Y., Asanuma, K. & Yanagisawa, K. Hearing Res. 2, 431 (1980).
Dallos, P., Santos-Sacchi, J. & Flock, A. Science 218, 582–584 (1982).
Davis, H. Cold Spring Harb. Symp. quant. Biol. 30, 181–189 (1965).
Corey, D. P. & Hudspeth, A. J. Nature 281, 675–677 (1979).
Evans, E. F. Archs Otolar. 105, 185–186 (1979).
Russell, I. J. & Sellick, P. M. J. Physiol., Lond. 284, 261–290 (1978).
Crawford, A. C. & Fettiplace, R. J. Physiol., Lond. 315, 317–338 (1981).
Geisler, C. D., Mountain, D. C., Hubbard, A. E., Adrian, H. O. & Ravindran, A. J. acoust. Soc. Am. 61, 1557–1566 (1977).
Durrant, J. D. & Gans, D. Acta oto-lar. 80, 13–18 (1975).
Mountain, D. C., Hubbard, A. E. and Geisler, C. D. Hearing Res. 3, 215–229 (1980).
Mountain, D. C. Science 210, 71–72 (1980).
Siegel, J. H. & Kim, D. O. Hearing Res. 6, 171–182 (1982).
Neher, E. & Stevens, C. F. A. Rev. Biophys. Bioenng 6, 345–381 (1977).
Hudspeth, A. J. J. Neurosci. 2, 1–10 (1982).
Ashmore, J. F. & Russell, I. J. Bioacoustics. (ed Lewis. B.) (Academic, London, in the press).
Engstrom, H., Ades, H. W. & Hawkins, J. E. J. acoust. Soc. Am. 34, 1356–1363 (1962).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Russell, I. Origin of the receptor potential in inner hair cells of the mammalian cochlea—evidence for Davis' theory. Nature 301, 334–336 (1983). https://doi.org/10.1038/301334a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/301334a0
This article is cited by
-
The origin of mechanical harmonic distortion within the organ of Corti in living gerbil cochleae
Communications Biology (2021)
-
Two-tone distortion in reticular lamina vibration of the living cochlea
Communications Biology (2020)
-
An audio attention computational model based on information entropy of two channels and exponential moving average
Human-centric Computing and Information Sciences (2019)
-
A connexin30 mutation rescues hearing and reveals roles for gap junctions in cochlear amplification and micromechanics
Nature Communications (2017)
-
Classifying Human Audiometric Phenotypes of Age-Related Hearing Loss from Animal Models
Journal of the Association for Research in Otolaryngology (2013)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.