Abstract
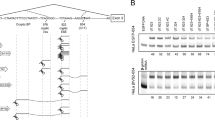
As is typical of all β-thalassaemias1, the erythroid cells of individuals with the variant haemoglobin E (α2β226Glu→Lys) exhibit a quantitative deficiency in their content of β-globin (in this case βE-globin) and its messenger RNA2,3. To determine the molecular basis of this phenotype, we have investigated the structure and expression of cloned βE-globin genes. We report here that the complete nucleotide sequence of a βE-gene revealed the expected GAG → AAG change in codon 26 but no other mutations. Expression of βE-globin genes introduced into HeLa cells revealed two abnormalities of RNA processing: slow excision of intervening sequence-1 (IVS-1) and alternative splicing into exon-1 at a cryptic donor sequence within which the codon 26 nucleotide substitution resides. These results demonstrate a disturbance in the expression of the βE-gene attributable solely to the exon mutation—a novel mechanism for gene dysfunction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Weatherall, D. J. & Clegg, J. B. Cell 29, 7–9 (1982).
Traeger, J., Wood, W. G., Clegg, J. B. & Weatherall, D. J. Nature 288, 497–499 (1980).
Benz, E. J. Jr et al. J. clin. Invest. 68, 118–126 (1981).
Antonarakis, S. E. et al. Proc. natn. Acad. Sci. U.S.A. 79, 6608–6611 (1982).
Maxam, A. & Gilbert, W. Meth. Enzym. 65, 499–560 (1980).
Treisman, R., Proudfoot, N. J., Shander, M. & Maniatis, T. Cell 29, 903–911 (1982).
Banerji, J., Rusconi, S. & Schaffner, W. Cell 27, 299–308 (1981).
Busslinger, M., Moschonas, N. & Flavell, R. A. Cell 27, 289–308 (1981).
Thomas, P. Proc. natn. Acad. Sci. U.S.A. 77, 5201–5205 (1980).
Weaver, R. F. & Weissmann, C. Nucleic Acids Res. 7, 1175–1193 (1979).
Breathnach, R. & Chambon, P. A. Rev. Biochem. 50, 349–383 (1981).
Mount, S. M. Nucleic Acids Res. 10, 459–472 (1982).
Felber, B. K., Orkin, S. H. & Hamer, D. H. Cell 29, 895–902 (1982).
Fukumaki, Y. et al. Cell 28, 585–593 (1982).
Traeger, J., Winichagoon, P. & Wood, W. H. J. clin. Invest. 69, 1050–1053 (1982).
Orkin, S. H. et al. Nature 296, 627–631 (1982).
Favaloro, J., Treisman, R. & Kamen, R. Meth. Enzym. 68, 718–749 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Orkin, S., Kazazian, H., Antonarakis, S. et al. Abnormal RNA processing due to the exon mutation of βE-globin gene. Nature 300, 768–769 (1982). https://doi.org/10.1038/300768a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/300768a0
This article is cited by
-
A novel SNP rs11759328 on Rho GTPase-activating protein 18 gene is associated with the expression of Hb F in hemoglobin E-related disorders
Annals of Hematology (2020)
-
Genetic determinants related to pharmacological induction of foetal haemoglobin in transfusion-dependent HbE-β thalassaemia
Annals of Hematology (2019)
-
Germline mutations in ETV6 are associated with thrombocytopenia, red cell macrocytosis and predisposition to lymphoblastic leukemia
Nature Genetics (2015)
-
Descriptive profile of β-thalassemia mutations in West Bengal population: a hospital-based study
International Journal of Hematology (2014)
-
Relationships of mRNA-protein secondary structures in the human β-globin gene HBB and four variants
Chinese Science Bulletin (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.