Abstract
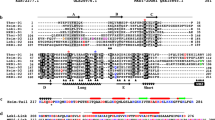
The actins constitute a family of highly conserved proteins found in all eukaryotic cells. Their conservation through a very wide range of taxonomic groups and the existence of tissue-specific isoforms make the actin genes very interesting for the study of the evolution of genes and their controlling elements. On the basis of amino acid sequence data, at least six different mammalian actins have been identified (skeletal muscle, cardiac muscle, two smooth muscle actins and the cytoplasmic β- and γ-actins)1–5. Rat spleen DNA digested by the EcoRI restriction enzyme contains at least 12 different fragments with actin-like sequences but only one which hybridized, in very stringent conditions, with the skeletal muscle cloned cDNA probe6. Here we describe the sequence of the actin gene in that fragment. The nucleotide sequence codes for two amino acids, Met-Cys, preceding the known N-terminal Asp of the mature protein. There are five small introns in the coding region and a large intron in the 5′-untranslated region. Comparison of the structure of the rat skeletal muscle actin gene with the available data on actin genes from other organisms shows that while the sequenced actin genes from Drosophila and yeast have introns at different locations, introns located at codons specifying amino acids 41, 121, 204 and 267 have been preserved at least from the echinoderm to the vertebrates. A similar analysis has been done by Davidson7. An intron at codon 150 is common to a plant actin gene and the skeletal muscle acting gene.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Storti, R. V. & Rich, A. Proc. natn. Acad. Sci. U.S.A. 73, 2346–2350 (1976).
Whalen, R. G., Butler-Browne, G. S. & Gros, F. Proc. natn. Acad. Sci. U.S.A. 73, 2018–2022 (1976).
Garrels, J. & Gibson, W. Cell 9, 793–805 (1976).
Elzinga, M. & Lu, R. in Contractile Systems in Nonmuscle Tissues (eds Perry, S. V., Margreth, A. & Adelstein, R.) 29 (North-Holland, Amsterdam, 1976).
Vanderkerchkove, J. & Weber, K. Differentiation 14, 124–133 (1979).
Nudel, U. et al. Proc. natn. Acad. Sci. U.S.A. 79, 2763–2767 (1982).
Davidson, E. H. in Genome Evolution (eds Dover, G. A. & Flavell, R. B.) (Academic, New York, 1982).
Fyrberg, E. A., Bond, B. J., Hershel, N. D., Mixter, K. S. & Davidson, N. Cell 24, 107–116 (1981).
Katcoff, D. et al. Proc. natn. Acad. Sci. U.S.A. 77, 960–964 (1980).
Shani, M. et al. Nucleic Acids Res. 9, 579–989 (1981).
Breathnach, R. & Chambon, P. A. Rev. Biochem. 50, 349–383 (1981).
Weaver, R. F. & Weissmann, C. Nucleic Acids Res. 6, 1175–1193 (1979).
Goldberg, M. thesis, Stanford Univ. (1979).
Ziff, E. B. & Evans, R. M. Cell 15, 1463–1475 (1978).
Yaffe, D. et al. in Stability and Switching in Cell Differentiation (eds Clayton, R. N. & Truman, D. E. S.) (Plenum, New York, in the press).
Gilbert, W. R. Nature 271, 501 (1978).
Shah, D. M., Hightower, R. C. & Mengher, R. B. Proc. natn. Acad. Sci. U.S.A. 72, 1022–1026 (1982).
Firtel, R. A., Timm, R., Kimmel, A. R. & McKeown, M. Proc. natn. Acad. Sci. U.S.A. 76, 6206–6210 (1979).
Gallwitz, D. & Sures, I. Proc. natn. Acad. Sci. U.S.A. 77, 2546–2550 (1980).
Ng, R. & Abelson, J. Proc. natn. Acad. Sci. U.S.A. 77, 3912–3916 (1980).
Maxam, A. M. & Gilbert, W. R. Proc. natn. Acad. Sci. U.S.A. 74, 560–564 (1977).
Sanger, F., Nicklen, S. & Coulson, A. R. Proc. natn. Acad. Sci. U.S.A. 74, 5463–5468 (1977).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zakut, R., Shani, M., Givol, D. et al. Nucleotide sequence of the rat skeletal muscle actin gene. Nature 298, 857–859 (1982). https://doi.org/10.1038/298857a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/298857a0
This article is cited by
-
In remembrance of David Yaffe
Skeletal Muscle (2020)
-
Phylogenetic analysis of NAP, an unconventional actin of the Volvocales
Plant Systematics and Evolution (2015)
-
A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD
Nature (1998)
-
Contractile protein gene expression in serum-free cultured adult rat cardiac myocytes
Pfl�gers Archiv European Journal of Physiology (1993)
-
Structural characterization of a rice actin gene
Plant Molecular Biology (1990)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.