Abstract
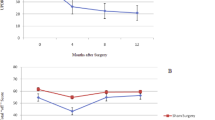
Rats with unilateral lesions of substantia nigra pars compacta (SN), the area of the brain containing most dopamine-containing neurones, are a widely recognized animal model of Parkinson's disease1–4. When given dopamine agonists such as apomorphine, such rats rotate in a direction contralateral to the lesion, presumably because of the development of supersensitive dopamine receptors in the striatum ipsilateral to the lesion. When grafts of embryonic SN are placed in the lateral ventricle5,6, or into a transplant cavity7 adjacent to the striatum in animals with SN lesions, this rotational behaviour has been shown to decrease. Histochemical examinations have shown that axons from the grafts have grown into the striatum5–7, and biochemical measurements indicate that dopamine concentrations are increased in areas of the striatum adjacent to the SN grafts6. Nevertheless, an obvious problem with this technique, both for basic research and possible clinical applications, is the requirement for fetal central nervous donor tissue. We now describe how grafts of adrenal medulla can be used with similar effects, involving chromaffin cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Andén, N. E. in Advances in Parkinsonism (eds Birkmayer, W. & Hornikiewicz, O.) 169–177 (Hoffman-LaRoche & Co. Ltd, Basel, 1975).
Ungerstedt, U. in The Neurosciences Third Study Program (eds Schmitt, F. O. & Worden, F. G.) 695–703. (MIT Press, Cambridge, 1974).
Glick, S. D., Jerussi, T. P. & Fleisher, L. N. Life Sci. 18, 889–896 (1976).
Costall, B., Naylor, R. J. & Pycock, C. J. Pharm. Pharmac. 27, 943–946 (1975).
Perlow, M. J. et al. Science 204, 643–647 (1979).
Freed, W. J. et al. Ann. Neural 8, 510–519 (1980).
Bjorklund, A., Dunnett, S. B., Stenevi, U., Lewis, M. E. & Iverson, C. D. Brain Res. 199, 307–333 (1980).
Molinoff, P. B. & Axelrod, J. A. Rev. Biochem. 40, 465–500 (1971).
Blaschko, H. & Muscholl, E. Handb. Exp. Pharmac. 33, (1972).
Olson, L. Histochemie 22, 1–7 (1970).
Olson, L., Seiger, A., Ebendahl, T. & Hoffer, B. Adv. Biochem. Pharmac. 25, 27–34 (1980).
Unsiker, K., Tschechne, B. & Tschechne, D. Brain Res. 152, 334–340 (1978).
Unsicker, K., Krisch, B., Otten, U. & Thoenen, H. Proc natn. Acad. Sci. U.S.A. 75, 3498–3502 (1978).
Unsicker, K. & Chamley, J. H., Tissue Res. 177, 247–268 (1977).
Unsicker, K., Rieffert, B. & Ziegler, W. Adv. Biochem. Pharmac., 25, 51–59 (1980).
Olson, L., Seiger, A., Freedman, R. & Hoffer, B. Expl Neurol. 70, 414–426 (1980).
Pellegrino, L. J. & Cushman, A. J. A Stereotaxic Atlas of the Rat Brain (Meredith, New York, 1967).
Corrodi, H. & Jonsson, G. J. Histochem. Cytochem. 15, 65–78 (1967).
Falck, B., Hillarp, N.-A., Thieme, G. & Torp, A. J. Histochem. Cytochem. 10, 348–354 (1962).
Olson, L. & Ungerstedt, U. Histochemie 22, 8–19 (1970).
Piezzi, R. S. & Cavicchia, J. C. Anat. Rec. 175, 77–86 (1973).
Wood, J. G. & Barnett, R. J. Anat. Rec. 145, 301–301 (1963).
Tischler, A. S. & Greene, L. A. Adv. Biochem Pharmac. 25, 61–68 (1980).
Ebendal, T., Olson, L., Seiger, A. & Hedlund, K. O. Nature 286, 25–28 (1980).
Perlow, M. J., Kumakura, K. & Guidotti, A. Proc. natn. Acad. Sci. U.S.A. 77, 5278–5281 (1980).
Olson, L., Ebendal, T. & Seiger, A. Devl Neurosci. 2, 160–176 (1979).
Freed, W. J. Brain Res. Bull. 1, 393–412 (1976).
Lundberg, J. M. et al. Proc. natn. Acad. Sci. U.S.A. 76, 4079–4083 (1979).
Yang, H.-Y. T., Hexum, T. & Costa, E. Life Sci. 27, 1119–1125 (1980).
Viveros, O. H., Dilberto, E. J., Hazum, E. & Chang, K.-J. Molec. Pharmac. 16, 1101–1108 (1979).
Greene, L. A. Adv. Pharmac. Ther. 10, 197–206 (1978).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Freed, W., Morihisa, J., Spoor, E. et al. Transplanted adrenal chromaffin cells in rat brain reduce lesion-induced rotational behaviour. Nature 292, 351–352 (1981). https://doi.org/10.1038/292351a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/292351a0
This article is cited by
-
The role of nonhuman primate models in the development of cell-based therapies for Parkinson’s disease
Journal of Neural Transmission (2018)
-
Genome-wide discovery of long intergenic noncoding RNAs and their epigenetic signatures in the rat
Scientific Reports (2017)
-
Regenerative medicine for Parkinson’s disease using differentiated nerve cells derived from human buccal fat pad stem cells
Human Cell (2017)
-
Cell-based therapies for Parkinson disease—past insights and future potential
Nature Reviews Neurology (2015)
-
Modulation of Dopaminergic Neuronal Differentiation from Sympathoadrenal Progenitors
Journal of Molecular Neuroscience (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.