Abstract
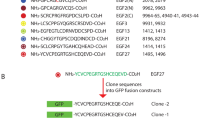
An amino acid sequence analysis of the N-terminal immunoglobulin heavy and light chain variable regions (VH and VL) from 16 hybridoma proteins which bind phosphorylcholine as well as the complete sequence analysis of 9 of these VH regions is presented. There seem to be more VH regions participating in the phosphorylcholine response than can be encoded directly by germ-line VH gene segments. Moreover, the V regions from IgG antibodies are considerably more variable than those from their IgM counterparts. These observations raise the possibility that a somatic mechanism for V region diversification produces greater diversity in IgG than in IgM antibodies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Weigert, M. & Riblet, R. Cold Spring Harb. Symp. quant. Biol. 41, 837–846 (1976).
Tonegawa, S., Hozumi, N., Mathyssens, G. & Schuller, R. Cold Spring Harb. Symp. quant. Biol. 41, 877–889 (1976).
Schilling, J., Clevinger, B., Davie, J. M. & Hood, L. Nature 283, 35–40 (1980).
Hood, L. et al. Cold Spring Harb. Symp. quant. Biol. (in the press).
Weigert, M., Gatmaitan, L., Loh, E., Schilling, J. & Hood, L. Nature 276, 785–790 (1978).
Max, E., Seidman, J. & Leder, P. Proc. natn. Acad. Sci. U.S.A. 76, 3450–3454 (1979).
Sakano, H., Hüppi, K., Heinrich, G. & Tonegawa, S. Nature 280, 288–294 (1979).
Early, P., Huang, H., Davis, M., Calame, K. & Hood, L. Cell 19, 981–992 (1980).
Wu, T. & Kabat, E. J. exp. Med. 132, 211–250 (1970).
Capra, J. D. & Kehoe, J. Proc. natn. Acad. Sci. U.S.A. 71, 845–848 (1974).
Amzel, L., Poljak, R., Saul, F., Varga, J. & Richard, F. Proc. natn. Acad. Sci. U.S.A. 71, 1427–1430 (1974).
Padlan, E., Segal, D., Spande, T., Davies, D., Rudikoff, S. & Potter, M. Nature new Biol. 245, 165–167 (1973).
Hood, L., Gray, W., Sanders, B. & Dreyer, W. Cold Spring Harb. Symp. quant. Biol. 32, 133–146 (1967).
Milstein, C. Nature 216, 330–332 (1967).
Potter, M. Adv. Immun. 25, 141–211 (1977).
Bernard, O., Hozumi, N. & Tonegawa, S. Cell 15, 1133–1144 (1978).
Seidman, J., Leder, A., Nau, M., Norman, B. & Leder, P. Science 202, 11–17 (1978).
Brack, C., Hirama, M., Lenhard-Schuller, R. & Tonegawa, S. Cell 15, 1–14 (1978).
Davis, M., Early, P., Calame, K., Livant, D. & Hood, L. ICN-UCLA Symposium on Eucaryotic Gene Regulation (eds Axel, R., Maniatis, T. & Fox, C. F.) 393–406 (Academic, New York, 1979).
Sakano, H., Maki, R., Kurosawa, Y., Roeder, W. & Tonegawa, S. Nature 286, 676–683 (1980).
Köhler, G. & Milstein, C. Nature 256, 495–497 (1975).
Hood, L. et al. Cold Spring Harb. Symp. quant. Biol. 41, 817–836 (1976).
Kluskens, L., Lee, W. & Köhler, H. Eur. J. Immun. 5, 489–496 (1975).
Rudikoff, S. & Claflin, J. L. J. exp. Med. 144, 1294–1304 (1976).
Sher, A. & Cohn, M. Eur. J. Immun. 2, 319–326 (1972).
Cosenza, H. & Köhler, H. Science 176, 1027–1029 (1972).
Sigal, N. H., Gearhart, P. J., Press, J. L. & Klinman, N. R. Nature 259, 51–52 (1976).
Lieberman, R., Potter, M., Mushinski, E. B., Humphrey, W. & Rudikoff, S. J. exp. Med. 139, 983–1001 (1974).
Gearhart, P. J. & Cebra, J. J. Nature 272, 264–265 (1978).
Strayer, D. S., Cosenza, H., Lee, W. M. F., Rowley, D. A. & Köhler, H. Science 186, 640–643 (1974).
Bottomly, K. & Mosier, D. E. J. exp. Med. 150, 1399–1409 (1979).
Gearhart, P. J. & Cebra, J. J. J. exp. Med. 149, 216–227 (1979).
Barstad, P. et al. Science, 183, 962–964 (1974).
Claflin, J. L. Eur. J. Immun. 6, 669–674 (1976).
Kabat, E. A., Wu, T. T. & Bilofsky, H. Sequences of Immunoglobulin Chains (US Department of Health, Education and Welfare, 1979).
Baltimore, D. Nature 248, 409–411 (1974).
Cohn, M. et al. ICN-UCLA Symposium on the Immune System: Genes, Receptors, Signals (eds Sercarz, E., Williamson, A. R. & Fox, C. F.) 89–117 (Academic, New York, 1974).
Cebra, J., Koo, P. & Ray, A. Science 186, 263–265 (1974).
Gally, J. A. & Edelman, G. A. Rev. Genet. 6, 1–46 (1972).
Reth, M., Hämmerling, G. J. & Rajewsky, K. Eur. J. Immun. 8, 393–400 (1978).
Marshak-Rothstein, A., Siekevitz, M., Margolies, M. N., Mudgett-Hunter, M. & Gefter, M. L. Proc. natn. Acad. Sci. U.S.A. 77, 1120–1124 (1980).
Estess, P., Lamoyi, E., Nisonoff, A. & Capra, J. D. J. exp. Med. 151, 863–875 (1980).
Early, P. W., Davis, M. M., Kaback, D. B., Davidson, N. & Hood, L. Proc. natn. Acad. Sci. U.S.A 76, 857–861 (1979).
Joho, R., Weissman, I. L., Early, P., Cole, J. & Hood, L. Proc. natn. Acad. Sci. U.S.A. 77, 1106–1110 (1980).
Klein, J. Biology of the Mouse Histocompatibility-2 Complex (Springer, New York, 1975).
Potter, M. Cell 21, 589–590 (1980).
Raff, M. C., Megson, M., Owen, J. J. T. & Cooper, M. D. Nature 259, 224–226 (1976).
Davis, M. et al. Nature 283, 733–739 (1980).
Kataoka, T., Kawakami, T., Takahashi, N. & Honjo, T. Proc. natn. Acad. Sci. U.S.A. 77, 919–923 (1980).
Brenner, S. & Milstein, C. Nature 211, 242–244 (1966).
Rodwell, J. D. & Karush, F. Molec. Immun. 17, 1553–1561 (1980).
Karush, F. Comprehensive Immunology, Vol. 5 (eds Litman, G. & Good, R.) 85–116 (Plenum, New York, 1978).
Gearhart, P. J., Sigal, N. H. & Klinman, N. R. J. exp. Med. 141, 56–71 (1975).
Shulman, M., Wilde, C. D. & Köhler, G. Nature 276, 269–270 (1978).
Kennett, R. H., Denis, K. A., Tung, A. S. & Klinman, N. R. Curr. Topics Microbiol. Immun. 81, 77–91 (1978).
Hurwitz, J., Coleclough, C. & Cebra, J. Cell 22, 349–359 (1980).
Chesebro, B. & Metzger, H. Biochemistry 11, 766–771 (1972).
Johnson, N. D., Hunkapiller, M. W. & Hood, L. E. Analyt. Biochem. 100, 335–338 (1979).
Hunkapiller, M. W. & Hood, L. E. Biochemistry 17, 2124–2133 (1978).
Dayhoff, M. O. Atlas of Protein Sequence and Structure (National Biomedical Research Foundation, Silver Spring, Maryland, 1976).
Kocher, H. P., Berek, C., Schreier, M. H., Cosenza, H. & Jaton, J-C. Eur. J. Immun. 10, 264–267 (1980).
Barstad, P. et al. Eur. J. Immun. 8, 497–503 (1978).
Rudikoff, S. & Potter, M. Biochemistry 17, 2703–2707 (1978).
Rudikoff, S. & Potter, M. Biochemistry 13, 4033–4038 (1974).
Rudikoff, S. & Potter, M. Proc. natn. Acad. Sci. U.S.A. 73, 2109–2112 (1976).
Johnson, N. thesis, California Inst. Technol. (1980).
Scatchard, G. Ann. N.Y. Acad. Sci. 51, 660–672 (1949).
Karush, F. Adv. Immun. 2, 1–40 (1962).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gearhart, P., Johnson, N., Douglas, R. et al. IgG antibodies to phosphorylcholine exhibit more diversity than their IgM counterparts. Nature 291, 29–34 (1981). https://doi.org/10.1038/291029a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/291029a0
This article is cited by
-
Structural, functional, and physiological properties of anti-(4-hydroxy-3-nitrophenyl)acetyl antibodies during the course of affinity maturation
Biophysical Reviews (2022)
-
Noncoding RNA processing by DIS3 regulates chromosomal architecture and somatic hypermutation in B cells
Nature Genetics (2021)
-
Near-infrared light-activated cancer cell targeting and drug delivery with aptamer-modified nanostructures
Nano Research (2016)
-
Impaired clearance of apoptotic cells in germinal centers: implications for loss of B cell tolerance and induction of autoimmunity
Immunologic Research (2011)
-
Physical and biological properties of homophilic therapeutic antibodies
Cancer Immunology, Immunotherapy (2011)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.