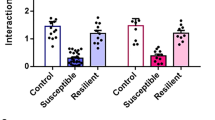
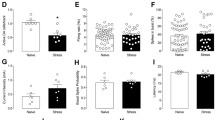
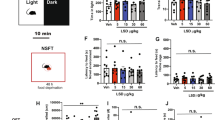
Abstract
Recent studies suggest that the mesocortico-frontal dopaminergic neurones which originate in the ventral tegmental area (VTA) have an inhibitory role in locomotor activity1,2. They are also markedly activated under stress3–5. This effect was shown in rats and mice subjected to electric foot-shocks by measuring either the rate of decline of dopamine (DA) after α-methylparatyrosine treatment3 or the changes in dihydroxyphenylacetic acid (dopac) levels and the dopac/DA ratio4–6. In rats, stress-induced activation of the dopaminergic neurones was prevented by benzodiazepines4,5, and studies in BALB/c mice introduced for 2 min into an open field further established the role of dopaminergic neurones in emotional responses7. These observations led us to examine the effects of long-term isolation on the activity of the mesocortico-frontal dopaminergic neurones in rats, some of which were subjected to a stressful situation. Indeed, several groups have reported that long-term isolation in rodents induced behavioural disturbances such as increased motor activity8,9 and aggression9,10 and hyper-reactivity to a new environment or stressful stimuli10,11. As measured by the changes in dopac levels or the dopac/DA ratio, we report here that the activity of the mesocortico-frontal dopaminergic neurones was reduced after isolation. This was not the case for the dopaminergic neurones projecting to the nucleus accumbens or the striatum, the rate of DA utilisation in these structures was even enhanced in isolated rats in which the activity of the mesocortico-frontal dopaminergic neurones was markedly reduced. Finally, we will show that a 3-min electric foot-shock session is more effective in enhancing dopac levels or the dopac/DA ratio in the frontal cortex of isolated than grouped rats.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Tassin, J. P. et al. Brain Res. 141, 267–281 (1978).
Carter, C. J. & Pycock, D. J. Br. J. Pharmac. 64, 430P (1978).
Thierry, A. M., Tassin, J. P., Blanc, G. & Glowinski, J. Nature 263, 242–244 (1976).
Lavielle, S. et al. Brain Res. 168, 585–594 (1979).
Fadda, F. et al. Life Sci. 27, 2219–2224 (1978).
Hervé, D. et al. Life Sci. 25, 1659–1664 (1979).
Tassin, J. P., Hervé D., Blanc, G. & Glowinski, J. Neurosci. Lett. (in the press).
Weinstock, M., Speiser, Z. & Ashkenazi, R. Psychopharmacology 56, 205–209 (1978).
Valzelli, L. Psychopharmacologia 31, 305–320 (1973).
Welch, A. S. & Welch, B. L. 91–142 (Plenum, New York, 1971).
Brain, P. & Benton, D. Life Sci. 24, 99–116 (1979).
Gauchy, C., Tassin, J. P., Glowinski, J. & Chéramy, A. J. Neurochem. 26, 99–104 (1976).
Sheving, L. E., Harrison, W. H., Gordon, N. P. & Pauly, J. E. Am. J. Physiol. 214, 166–173 (1968).
Thoa, N., Tizabi, Y. & Jacobowitz, D. Brain Res. 131, 259–269 (1977).
Lisoprawski, A., Hervé, D., Blanc, G., Glowinski, J. & Tassin, J. P. Brain Res. 183, 229–234 (1980).
Hervé, D. et al. Neurosci. Lett. 15, 127–133 (1979).
LeMoal, M. et al. Adv. biochem. Psychopharmac. 16, 237–245 (1977).
Brozoski, T. S., Brown, R. M., Rosvold, H. E. & Goldman, P. S. Science 205, 929 (1979).
Simon, H., Scatton, B. & Le Moal, M. Neurosci. Lett. 15, 319–324 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Blanc, G., Hervé, D., Simon, H. et al. Response to stress of mesocortico-frontal dopaminergic neurones in rats after long-term isolation. Nature 284, 265–267 (1980). https://doi.org/10.1038/284265a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/284265a0
This article is cited by
-
Behavioral and Noradrenergic Sensitizations in Vulnerable Traumatized Rats Suggest Common Bases with Substance Use Disorders
Molecular Neurobiology (2019)
-
Chronic stress induces steatohepatitis while decreases visceral fat mass in mice
BMC Gastroenterology (2014)
-
Changes in dopamine D2-receptor binding are associated to symptom reduction after psychotherapy in social anxiety disorder
Translational Psychiatry (2012)
-
Social isolation stress induces ATF-7 phosphorylation and impairs silencing of the 5-HT 5B receptor gene
The EMBO Journal (2010)
-
In rats chronically treated with clozapine, tyrosine depletion attenuates the clozapine-induced in vivo increase in prefrontal cortex dopamine and norepinephrine levels
Psychopharmacology (2006)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.