Abstract
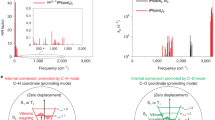
The photovoltaic responses of meso tetraphenyl-, meso tetra-propyl- and octaethyl-porphyrins, porphines, chlorins, cofacial diporphyrins and mesoporphyrin IX diesters were investigated using two cell configurations: Al| Porphyrin |Ag, and Al| Porphyrin | Fe(CN)6−3, Fe(CN)6−4 |Pt. We found (1) that the Al–porphyrin interface is photoactive: the action spectra closely follow the absorption spectra of the porphyrins, and this interface is best described as a semiconductor–insulator–metal diode consisting of porphyrin |A12O3 |Al; (2) that within a homologous series, in which the porphyrin skeleton is fixed but the metal is varied, the quantum yields parallel the ease of oxidation of the porphyrin in nonaqueous solvents. The more easily oxidised compounds exhibit the higher quantum yields; (3) no obvious correlations are found with the luminescent properties of the porphyrins in solution; (4) the morphology of the films influences the quantum yields: amorphous films are better than microcrystalline ones; and (5) the most efficient cells reach quantum yields of ∼0.2 and energy efficiencies of ∼1% for monochromatic light at the peak of the action spectrum in the region of 400–450 nm.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Putseiko, E. & Akimov, I. Discuss. Faraday Soc. 27, 83–93 (1959).
McCree, K. Biochim. biophys. Acta 102, 96–102 (1965).
Fedorov, M. & Benderskii, V. Sov. Phys.-Semicond. 4, 1198–1199, 1720–1722 (1971).
Lyons, L. & Newman, O. Aust. J. Chem. 24, 13–23 (1973).
Ghosh, A. & Feng, T. J. appl. Phys. 44, 2781–2788 (1973).
Ghosh, A., Morel, D., Feng, T., Shaw, R. & Rowe, C. J. appl. Phys. 45, 230–236 (1974).
Bromberg, A., Tang, C. & Albrecht, A. J. chem. Phys. 60, 4058–4062 (1974).
Tang, C. & Albrecht, A. Nature 254, 507–509 (1975).
Tang, C. & Albrecht, A. J. chem. Phys. 62, 2139–2149 (1975).
Tang, C. & Albrecht, A. J. chem. Phys. 63, 953–967 (1975).
Merritt, V. & Hovel, H. Appl. phys. lett. 29, 414–415 (1976).
Tsubomura, H., Matsumura, M., Nomura, Y. & Amamiya, T. Nature 261, 402–403 (1976).
Corker, G. & Lundstrom, I. J. appl. Phys. 49, 686–700 (1978).
Tien, H. Solar Energy 21, 291–295 (1978).
Wraight, C. & Clayton, R. Biochim. biophys. Acta 333, 246–260 (1973).
Fajer, J., Brune, D., Davis, M., Forman, A. & Spaulding, L. Proc. natn. Acad. Sci. U.S.A. 72, 4956–4960 (1975).
Fujita, I., Davis, M. & Fajer, J. J. Am. chem. Soc. 100, 6280–6282 (1978).
Fan, F.-R. & Faulkner, L. J. chem. Phys. 69, 3334–3340 (1978).
Fan, F.-R. & Faulkner, L. J. chem. Phys. 69, 3341–3349 (1978).
Kampas, F. & Gouterman, M. J. phys. Chem. 81, 690–695 (1977).
Wang, J. Proc. natn. Acad. Sci. U.S.A. 62, 653–660 (1969).
Green, M., King, F. & Shewchun, J. Solid-St. Electron. 17, 551–561 (1974).
Fajer, J. & Davis, M. S. in The Porphyrins (ed. Dolphin, D.) (Academic, New York, 1979). Vol. 4, Ch. 4.
Gouterman, M. in The Porphyrins Vol. 3, Ch. 1 (ed. Dolphin D.) (Academic, New York, 1978).
Chang, C. K. J. Heterocyclic Chem. 14, 1285 (1977); Adv. Chem. Ser. 173, 162 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kampas, F., Yamashita, K. & Fajer, J. Photoelectrochemical properties of metalloporphyrins. Nature 284, 40–42 (1980). https://doi.org/10.1038/284040a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/284040a0
This article is cited by
-
Structure and photoelectric properties of LB films of porphyrin bearing pyridinium with long-chain alkyl group
Science in China Series B: Chemistry (1998)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.