Abstract
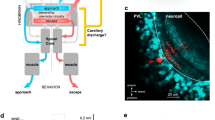
DURING rapid eye movements, saccades, human subjects do not perceive movement of their visual surroundings, and their sensitivity to light flashes and moving objects is reduced. This phenomenon is known as saccadic suppression. The physiological basis of this suppression is not well understood. The necessity for a mechanism which allows discrimination between retinal image movement generated by the object and that generated by the subject was recognised by Helmholtz1. Sperry2 proposed that along with the eye movement, the central nervous system generates a ‘corollary discharge’ into the visual centres that compensates for displacement of the retinal image, and von Hoist and Mittelstaedt3 independently postulated a similar ‘efference copy’ mechanism. Functionally significant efferent control has been demonstrated in several non-visual sensory pathways4,5. Such a mechanism could account for experimentally produced effects in human subjects6,7. The prevailing view8–12, however, is that saccadic suppression in higher mammals is caused not by corollary discharge but instead by inhibition derived from the retinal image motion generated by eye movement. This view has been extended to include saccadic suppression of the visual movement detector system in crickets and locusts13–15, in which the neural circuit performing the inhibitory function has been identified15,16. The experiments leading to this conclusion have all used passive movement of the animal with respect to the visual environment, or the reverse, and not true saccades. We have now investigated the effects of head movements by the locust on its visual responses, and report that movement detector neurones are strongly inhibited during the fast phase of optokinetic nystagmus and during voluntary sac-cades by a central nervous mechanism of the corollary discharge type. The effect of the inhibitory mechanisms of retinal origin are negligible by comparison.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Helmholtz, H. von Physiological Optics Vol. III (ed. Southall, J. P. C.) (Optical Society of America, Menasha, Wisconsin, 1925).
Sperry, R. W. J. comp. Physiol. Psych. 43, 482–489 (1950).
von Holst, E. & Mittelstaedt, H. Naturwissenschaften 37, 464–476 (1950).
Kuffler, S. W., Hunt, C. C. & Quilliam, J. P. J. Neurophysiol. 14, 29–54 (1951).
Flock, Å. & Russell, I. J. J. Physiol., Lond. 235, 591–605 (1973).
Teuber, H-L. in Handbook of Physiology, section I. Vol. 3 (ed. Field, J.) 1595–1668 (American Physiological Society, Washington, 1960).
Riggs, L. A., Merton, P. A. & Morton, H. B. Vision Res. 14, 997–1011 (1974).
Wurtz, R. H. J. Neurophysiol. 32, 975–986 (1969); 32, 987–994 (1969).
Noda, H. J. Physiol., Lond. 249, 87–102 (1975); 250, 579–595 (1975).
Judge, S. J. & Wurtz, R. H. Soc. Neurosci, 7th A. Meet. Abs. 3, 564 (1977).
Matin, E. Psychol. Bull. 81, 899–917 (1974).
Wurtz, R. H. & Campbell, F. W. Ass. Res. Vision Opthalmol., Spring Meeting, 1977, p. 106.
Palka, J. J. exp. Biol. 50, 723–732 (1969); Am. Zool. 12, 497–505 (1972).
Palka, J. J. Insect Physiol. 13, 235–238 (1967).
Rowell, C. H. F., O'Shea, M. & Williams, J. L. D. J. exp. Biol. 68, 157–185 (1977).
O'Shea, M. & Rowell, C. H. F. Nature 254, 53–55 (1975); J. exp. Biol. 65, 389–408 (1976).
Rowell, C. H. F. Z. vergl, Physiologie 73, 167–194 (1971).
Rowell, C. H. F. & O'Shea, M. J. exp. Biol. 65, 273–288 (1976).
Burrows, M. & Rowell, C. H. F. J. comp. Physiol. 85, 222–234 (1973).
Pearson, K. & Goodman, C. J. comp. Neurol. 184, 141–166 (1979).
Kien, J. & Land, M. F. Physiological Entomol. 3, 53–57 (1978).
Wallace, M., Blair, S. M. & Westheimer, G. Expl Brain Res. 33, 19–25 (1978).
Shepheard, P. J. exp. Biol. 60, 735–768 (1974).
Horn, G. & Rowell, C. H. F. J. exp. Biol. 49, 143–169 (1968).
Palka, J. Am. Zool. 7, 7–28 (1967).
Haskell, P. T. Nature 183, 1106–1107 (1959).
Goodman, L. J. J. exp. Biol. 42, 385–407 (1965).
Kien, J. J. comp. Physiol. 113, 161–180 (1977).
Collewijn, H. Vision Res. 9, 803–814 (1969).
Robinson, D. A. J. Physiol., Lond. 174, 245–264 (1964).
Robinson, D. L. & Wurtz, R. H. J. Neurophysiol. 39, 852–870 (1976).
Richmond, B. J. & Wurtz, R. H. Soc. Neurosci. 7th A. Meet. Abstr. 3, 574 (1977).
Benvenuto, L. A. & Rezak, M. Brain Res. 108, 1–24 (1976).
Sandeman, D. C. Comp. Biochem. Physiol. 24, 635–638 (1968).
Westheimer, G. Archs Ophthal. 52, 710–724 (1954).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ZARETSKY, M., ROWELL, C. Saccadic suppression by corollary discharge in the locust. Nature 280, 583–585 (1979). https://doi.org/10.1038/280583a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/280583a0
This article is cited by
-
Cellular evidence for efference copy in Drosophila visuomotor processing
Nature Neuroscience (2015)
-
Information flow through neural circuits for pheromone orientation
Nature Communications (2014)
-
Corollary discharge across the animal kingdom
Nature Reviews Neuroscience (2008)
-
Temperature-sensitive gating in a descending visual interneuron, DCMD
Journal of Comparative Physiology A (2006)
-
Corollary discharge inhibition and audition in the stridulating cricket
Journal of Comparative Physiology A (2005)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.