Abstract
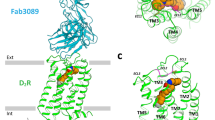
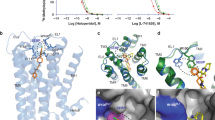
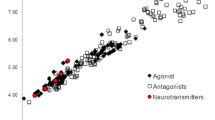
THE introduction of neuroleptics of the phenothiazine and thioxanthene classes has revolutionised the treatment of schizophrenia and there is increasing evidence1–3 that links this antipsychotic action to a blockade of dopamine receptors in the brain. The thioxanthene compounds are of particular interest as, because of the presence of an exocyclic double bond and a substituent in the tricyclic nucleus, they exist as cis–trans geometric isomers about the double bond with respect to the ring substituent (Fig. 1). Virtually all the neuroleptic activity is confined to the cis isomer4.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Horn, A. S., and Snyder, S. H., Proc. natn. Acad. Sci U.S.A., 68, 2325–2328 (1971).
Matthysse, S., Fedn Proc., 32, 200–205 (1973).
Snyder, S. H., Banerjee, S. P., Yamamura, H. I., and Greenburg, D., Science, 184, 1243–1253 (1974).
Moller-Nielsen, C. G., et al., Acta pharmac. tox., 33, 353–362 (1973).
Gottfries, C. G., Br. J. Psychiat., 119, 547–548 (1971).
Miller, R. J., Horn, A. S., and Iversen, L. L., Mol. Pharmac., 10, 759–766 (1974).
Horn, A. S., Post, M. L., and Kennard, O., J. Pharm. Pharmac. (in the press).
Miller, R. J., Horn, A. S., and Iversen, L. L., J. Pharm. Pharmac., 27, 212–213 (1975).
Bruderlein, F. T., Humber, L. G., and Voith, K., J. med. Chem., 18, 185–188 (1975).
York, D. H., Brain Res., 37, 91–99 (1972).
Clement-Cormier, Y. C., Kebebian, J. W., Petzold, G. L., and Greengard, P., Proc. natn. Acad. Sci. U.S.A., 71, 1113–1117 (1974).
Post, M. L., Kennard, O., and Horn, A. S., Acta crystallogr. B 30, 1644–1646 (1974).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
POST, M., KENNARD, O. & HORN, A. Stereoselective blockade of the dopamine receptor and the X-ray structures of α and β-flupenthixol. Nature 256, 342–343 (1975). https://doi.org/10.1038/256342a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/256342a0
This article is cited by
-
Pharmacologic circumvention of multidrug resistance
Cytotechnology (1993)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.