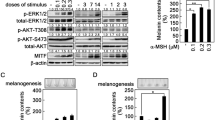
Abstract
TYROSINASE activity is greatly enhanced in cultured Cloudman S91 melanoma cells following addition of melanocyte-stimulating hormone (MSH) to the culture medium1. The increased activity occurs in the G2 phase of the cell cycle2 because membrane receptors for MSH are available only in this phase3. The response to MSH is mediated through cyclic AMP (refs 1–4). It is well documented that many peptide hormones function by activating membrane-bound adenyl cyclase molecules, causing net increases in intracellular cyclic AMP concentrations. These increases in turn have profound effects on cell division, morphology, and the expression of differentiated functions in a wide variety of cells and tissues. Little is known, however, about the levels at which genetic expression is regulated or the molecular intermediates involved in the regulation. For example, it is generally assumed that cyclic AMP-dependent protein kinases are involved in many of the responses because most tissues contain such enzymes5. The regulation of glycogenolysis is the best known example supporting this assumption6–7. But it cannot be taken as fact that in eukaryotes all cyclic AMP-mediated processes are post-translational in nature.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Pawelek, J., Wong, G., Sansone, M., and Morowitz, J., Yale J. Biol. Med., 46, 430–433 (1973).
Wong, G., Pawelek, J., Sansone, M., and Morowitz, J., Nature, 248, 351–354 (1974).
Varga, J. M., DiPasquale, A., Pawelek, J., McGuire, J. S., and Lerner, A. B., Proc. natn. Acad. Sci. U.S.A., 71, 1590–1593 (1974).
Bitensky, M. W., and Demopoulos, H. B., J. Invest. Dermat., 54, 83 (1970).
Kuo, J. F., and Greengard, P., Proc. natn. Acad. Sci. U.S.A., 64, 1349–1355 (1969).
Rall, T. W., and Sutherland, E. W., Cold Spring Harb. Symp. quant. Biol., 26, 347–354 (1961).
Krebs, E. G., Graves, D. J., and Fischer, E. H., J. Biol. Chem., 234, 378–384 (1960).
Pomerantz, S., Science, 164, 838–840 (1969).
Axen, R., Porath, J., and Ernback, S., Nature, 214, 1302–1304 (1967).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
WONG, G., PAWELEK, J. Melanocyte-stimulating hormone promotes activation of pre-existing tyrosinase molecules in Cloudman S91 melanoma cells. Nature 255, 644–646 (1975). https://doi.org/10.1038/255644a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/255644a0
This article is cited by
-
Phenylethanoid and phenylpropanoid glycosides with melanogenesis inhibitory activity from the flowers of Narcissus tazetta var. chinensis
Journal of Natural Medicines (2016)
-
A novel culture system to induce melanin synthesis by three-dimensional spheroid culture
Biotechnology and Bioprocess Engineering (2015)
-
Dimeric pyrrolidinoindoline-type alkaloids with melanogenesis inhibitory activity in flower buds of Chimonanthus praecox
Journal of Natural Medicines (2014)
-
Aromatic-turmerone inhibits α-MSH and IBMX-induced melanogenesis by inactivating CREB and MITF signaling pathways
Archives of Dermatological Research (2011)
-
Whitening activity of luteolin related to the inhibition of cAMP pathway in α-MSH-stimulated B16 melanoma cells
Archives of Pharmacal Research (2008)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.