Abstract
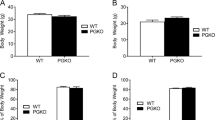
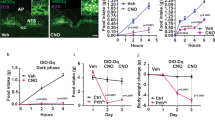
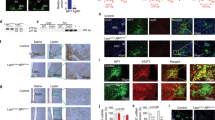
Feeding is influenced by hypothalamic neuropeptides that promote (orexigenic peptides) or inhibit feeding1. Of these, neuropeptide Y (NPY) in the arcuate nucleus2 and melanin-concentrating hormone (MCH)3 and orexins/hypocretins4,5 in the lateral hypothalamus have received attention because their expression is increased during fasting and because they promote feeding when administered centrally. Surprisingly, absence of the orexigenic neuropeptide NPY fails to alter feeding or body weight in normal mice6. As deficiency of a single component of the pathway that limits food intake (such as leptin or receptors for melanocortin-4)7,8 causes obesity, it has been suggested that orexigenic signals are more redundant than those limiting food intake7,8. To define further the physiological role of MCH and to test the redundancy of orexigenic signals, we generated mice carrying a targeted deletion of the MCH gene. MCH-deficient mice have reduced body weight and leanness due to hypophagia (reduced feeding) and an inappropriately increased metabolic rate, despite their reduced amounts of both leptin and arcuate nucleus pro-opiomelanocortin messenger RNA. Our results show that MCH is a critical regulator of feeding and energy balance which acts downstream of leptin and the melanocortin system, and that deletion of a gene encoding a single orexigenic peptide can result in leanness.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Flier, J. S. & Maratos-Flier, E. Obesity and the hypothalamus: novel peptides for new pathways. Cell 92, 437–440 (1998).
Dryden, S., Frankish, H., Wang, Q. & Williams, G. Neuropeptide Y and energy balance: one way ahead for the treatment of obesity? Eur. J. Clin. Invest. 24, 293–308 (1994).
Qu, D.et al. Arole for melanin-concentrating hormone in the central regulation of feeding behaviour. Nature 380, 243–247 (1996).
Sakurai, T.et al. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell 92, 573–585 (1998).
de Lecea, L.et al. The hypocretins: hypothalamus-specific peptides with neuroexcitatory activity. Proc. Natl Acad. Sci. USA 95, 322–327 (1998).
Erickson, J., Clegg, K. & Palmiter, R. Sensitivity to leptin and susceptibility to seizures of mice lacking neuropeptide Y. Nature 381, 415–418 (1996).
Zhang, Y.et al. Positional cloning of the mouse ob gene and its human homologue. Nature 372, 425–432 (1994).
Huszar, D.et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131–141 (1997).
Krezel, W.et al. Impaired locomotion and dopamine signaling in retinoid receptor mutant mice. Science 279, 863–867 (1998).
Ahima, R. S., Prabakaran, D. & Flier, J. S. Postnatal leptin surge and regulation of circadian rhythm of leptin by feeding. Implications for energy homeostasis and neuroendocrine function. J. Clin. Invest. 101, 1020–1027 (1998).
Halaas, J.et al. Weight reducing effect of the plasma protein encoded by the obese gene. Science 269, 543–546 (1995).
Ollmann, M. M.et al. Antagonism of central melanocortin receptors in vitro and in vivo by Agouti-related protein. Science 278, 135–138 (1997).
Shutter, J. R.et al. Hypothalamic expression of ART, a novel gene related to agouti, is up-regulated in obese and diabetic mutant mice. Genes Dev. 11, 593–602 (1997).
Tsujii, S. & Bray, G. A. Acetylation alters the feeding response to MSH and beta-endorphin. Brain Res. Bull. 23, 165–169 (1989).
Fan, W., Boston, B. A., Kesterson, R. A., Hruby, V. J. & Cone, R. D. Role of melanocortinergic neurons in feeding and the agouti obesity syndrome. Nature 385, 165–168 (1997).
Thornton, J. E., Cheung, C. C., Clifton, D. K. & Steiner, R. A. Regulation of hypothalamic roopiomelanocortin mRNA by leptin in ob/ob mice. Endocrinology 138, 5063–5066 (1997).
Mizuno, T. M.et al. Hypothalamic pro-opiomelanocortin mRNA is reduced by fasting in ob/ob and db/db mice, but is stimulated by leptin. Diabetes 47, 294–297 (1998).
Ahima, R. S.et al. Role of leptin in the neuroendocrine response to fasting. Nature 382, 250–252 (1996).
Bittencourt, J. C & Elias, C. F. Diencephalic origins of melanin-concentrating hormone immunoreactive projections to medial septum/diagonal band complex and spinal cord using two retrograde fluorescent tracers. Ann. NY Acad. Sci. 680, 462–465 (1993).
Elias, C. F.et al. Chemically defined projections linking the mediobasal hypothalamus and the lateral hypothalamic area. J. Comp. Neurol.(in the press).
Krude, H.et al. Severe early-onset obesity, adrenal insufficiency and red hair pigmentation caused by POMC mutations in humans. Nature Genet. 19, 155–157 (1998).
Nahon, J. L. The melanin-concentrating hormone: from the peptide to the gene. Crit. Rev. Neurobiol. 8, 221–262 (1994).
Rossi, M.et al. Melanin-concentrating hormone acutely stimulates feeding, but chronic administration has no effect on body weight. Endocrinology 138, 351–355 (1997).
Bittencourt, J. C.et al. The melanin-concentrating hormone system of the rat brain: an immuno- and hybridization histochemical characterization. J. Comp. Neurol. 319, 218–245 (1992).
Montague, C. T.et al. Congenital leptin deficiency is associated with severe early-onset obesity in humans. Nature 387, 903–908 (1997).
Breton, C., Presse, F., Hervieu, G. & Nahon, J. L. Structure and regulation of the mouse melanin concentrating hormone mRNA and gene. Mol. Cell. Neurosci. 4, 271–284 (1993).
Li, E., Sucov, H. M., Lee, K. F., Evans, R. M. & Jaenisch, R. Normal development and growth of mice carrying a targeted disruption of the alpha 1 retinoic acid receptor gene. Proc. Natl Acad. Sci. USA 90, 1590–1594 (1993).
Marks, D. L.et al. Simultaneous visualization of two cellular mRNA species in individual neurons by use of a new double in situ hybridization method. Mol. Cell. Neurosci. 3, 395–405 (1993).
Lowell, B. B.et al. Development of obesity in transgenic mice after genetic ablation of brown adipose tissue. Nature 366, 740–742 (1993).
Acknowledgements
We thank J. Mastaitis, C. Behn and C. Lee for technical assistance, J. Elmquist for help in analysing and interpreting brain anatomy, and D. S. Ludwig for the P1 clone used in making the construct for the knockout mice. This work was supported in part by grants from NIH to J.S.F. and E.M.-F., from the American Diabetes Association to E.M.-F., and from Eli Lilly to J.S.F. and E.M.-F., and by the Transgenic Core of the Boston Obesity Nutrition Research Center. M.S. was supported by the Banyu Fellowship in Lipid Metabolism and Atherosclerosis which is sponsored by Banyu Pharmaceutical Co Ltd and the Merck Foundation.
Author information
Authors and Affiliations
Author notes
Eleftheria Maratos-Flier: Correspondence and requests for materials should be addressed to E.M.-F
- Eleftheria Maratos-Flier
Rights and permissions
About this article
Cite this article
Shimada, M., Tritos, N., Lowell, B. et al. Mice lacking melanin-concentrating hormone are hypophagic and lean. Nature 396, 670–674 (1998). https://doi.org/10.1038/25341
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/25341
This article is cited by
-
Loss of hypothalamic MCH decreases food intake in amyotrophic lateral sclerosis
Acta Neuropathologica (2023)
-
Hypothalamic melanin-concentrating hormone regulates hippocampus-dorsolateral septum activity
Nature Neuroscience (2022)
-
Multifaceted actions of melanin-concentrating hormone on mammalian energy homeostasis
Nature Reviews Endocrinology (2021)
-
Involvement of MCH-oxytocin neural relay within the hypothalamus in murine nursing behavior
Scientific Reports (2021)
-
Sleep-Wake Control by Melanin-Concentrating Hormone (MCH) Neurons: a Review of Recent Findings
Current Neurology and Neuroscience Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.