Abstract
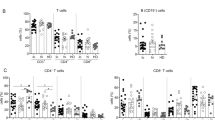
A link between chronic hepatitis C virus (HCV) infection and low-grade B-cell lymphomas has been suggested by epidemiological studies. Marginal zone lymphomas (MZLs) including splenic lymphomas with villous lymphocytes are among the most frequently reported subgroups in the setting of chronic HCV infection. In this study, we examined the effect of antiviral treatment in eight patients with HCV-associated MZL. We found that five out of eight patients have responded to interferon alpha and ribavirin. In some cases, hematologic responses were correlated to virologic responses. In addition, we report a case of large granular lymphocyte leukemia occurring in association with MZL and HCV, and responding to interferon and ribavirin. We suggest that there is an etiologic link between HCV and antigen-driven lymphoproliferative disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ferri C, Caracciolo F, Zignego AL, La Civita L, Monti M, Longombardo G et al. Hepatitis C virus infection in patients with non-Hodgkin's lymphoma. Br J Haematol 1994; 88: 392–394.
Luppi M, Longo G, Ferrari MG, Ferrara L, Marasca R, Barozzi P et al. Additional neoplasms and HCV infection in low-grade lymphoma of MALT type. Br J Haematol 1996; 94: 373–375.
Luppi M, Grazia Ferrari M, Bonaccorsi G, Longo G, Narni F, Barozzi P et al. Hepatitis C virus infection in subsets of neoplastic lymphoproliferations not associated with cryoglobulinemia. Leukemia 1996; 10: 351–355.
Mazzaro C, Zagonel V, Monfardini S, Tulissi P, Pussini E, Fanni M et al. Hepatitis C virus and non-Hodgkin's lymphomas. Br J Haematol 1996; 94: 544–550.
Silvestri F, Pipan C, Barillari G, Zaja F, Fanin R, Infanti L et al. Prevalence of hepatitis C virus infection in patients with lymphoproliferative disorders. Blood 1996; 87: 4296–4301.
Zignego AL, Ferri C, Giannini C, La Civita L, Careccia G, Longombardo G et al. Hepatitis C virus infection in mixed cryoglobulinemia and B-cell non-Hodgkin's lymphoma: evidence for a pathogenetic role. Arch Virol 1997; 142: 545–555.
King PD, Wilkes JD, Diaz-Arias AA . Hepatitis C virus infection in non-Hodgkin's lymphoma. Clin Lab Haematol 1998; 20: 107–110.
Collier JD, Zanke B, Moore M, Kessler G, Krajden M, Shepherd F et al. No association between hepatitis C and B-cell lymphoma. Hepatology 1999; 29: 1259–1261.
McColl MD, Singer IO, Tait RC, McNeil IR, Cumming RL, Hogg RB . The role of hepatitis C virus in the aetiology of non-Hodgkins lymphoma – a regional association? Leuk Lymphoma 1997; 26: 127–130.
Ellenrieder V, Weidenbach H, Frickhofen N, Michel D, Prummer O, Klatt S et al. HCV and HGV in B-cell non-Hodgkin's lymphoma. J Hepatol 1998; 28: 34–39.
Germanidis G, Haioun C, Pourquier J, Gaulard P, Pawlotsky JM, Dhumeaux D et al. Hepatitis C virus infection in patients with overt B-cell non-Hodgkin's lymphoma in a French center. Blood 1999; 93: 1778–1779.
Hausfater P, Cacoub P, Sterkers Y, Thibault V, Amoura Z, Nguyen L et al. Hepatitis C virus infection and lymphoproliferative diseases: prospective study on 1576 patients in France. Am J Hematol 2001; 67: 168–171.
Zuckerman E, Zuckerman T, Levine AM, Douer D, Gutekunst K, Mizokami M et al. Hepatitis C virus infection in patients with B-cell non-Hodgkin lymphoma. Ann Intern Med 1997; 127: 423–428.
Izumi T, Sasaki R, Tsunoda S, Akutsu M, Okamoto H, Miura Y . B cell malignancy and hepatitis C virus infection. Leukemia 1997; 11 (Suppl. 3): 516–518.
Rabkin CS, Tess BH, Christianson RE, Wright WE, Waters DJ, Alter HJ et al. Prospective study of hepatitis C viral infection as a risk factor for subsequent B-cell neoplasia. Blood 2002; 99: 4240–4242.
El-Serag HB, Hampel H, Yeh C, Rabeneck L . Extrahepatic manifestations of hepatitis C among United States male veterans. Hepatology 2002; 36: 1439–1445.
Mele A, Pulsoni A, Bianco E, Musto P, Szklo A, Sanpaolo MG et al. Hepatitis C virus and B-cell non-Hodgkin lymphomas: an Italian multicenter case–control study. Blood 2003; 102: 996–999.
Arcaini L, Paulli M, Boveri E, Vallisa D, Bernuzzi P, Orlandi E et al. Splenic and nodal marginal zone lymphomas are indolent disorders at high hepatitis C virus seroprevalence with distinct presenting features but similar morphologic and phenotypic profiles. Cancer 2004; 100: 107–115.
Zuckerman E, Zuckerman T, Sahar D, Streichman S, Attias D, Sabo E et al. bcl-2 and immunoglobulin gene rearrangement in patients with hepatitis C virus infection. Br J Haematol 2001; 112: 364–369.
Zignego AL, Ferri C, Giannelli F, Giannini C, Caini P, Monti M et al. Prevalence of bcl-2 rearrangement in patients with hepatitis C virus-related mixed cryoglobulinemia with or without B-cell lymphomas. Ann Intern Med 2002; 137: 571–580.
Sansonno D, De Vita S, Iacobelli AR, Cornacchiulo V, Boiocchi M, Dammacco F . Clonal analysis of intrahepatic B cells from HCV-infected patients with and without mixed cryoglobulinemia. J Immunol 1998; 160: 3594–3601.
Chan CH, Hadlock KG, Foung SK, Levy S . V(H)1–69 gene is preferentially used by hepatitis C virus-associated B cell lymphomas and by normal B cells responding to the E2 viral antigen. Blood 2001; 97: 1023–1026.
De Re V, De Vita S, Marzotto A, Rupolo M, Gloghini A, Pivetta B et al. Sequence analysis of the immunoglobulin antigen receptor of hepatitis C virus-associated non-Hodgkin lymphomas suggests that the malignant cells are derived from the rheumatoid factor-producing cells that occur mainly in type II cryoglobulinemia. Blood 2000; 96: 3578–3584.
Ivanovski M, Silvestri F, Pozzato G, Anand S, Mazzaro C, Burrone OR et al. Somatic hypermutation, clonal diversity, and preferential expression of the VH 51p1/VL kv325 immunoglobulin gene combination in hepatitis C virus-associated immunocytomas. Blood 1998; 91: 2433–2442.
Marasca R, Vaccari P, Luppi M, Zucchini P, Castelli I, Barozzi P et al. Immunoglobulin gene mutations and frequent use of VH1-69 and VH4-34 segments in hepatitis C virus-positive and hepatitis C virus-negative nodal marginal zone B-cell lymphoma. Am J Pathol 2001; 159: 253–261.
Quinn ER, Chan CH, Hadlock KG, Foung SK, Flint M, Levy S . The B-cell receptor of a hepatitis C virus (HCV)-associated non-Hodgkin lymphoma binds the viral E2 envelope protein, implicating HCV in lymphomagenesis. Blood 2001; 98: 3745–3749.
De Re V, De Vita S, Marzotto A, Gloghini A, Pivetta B, Gasparotto D et al. Pre-malignant and malignant lymphoproliferations in an HCV-infected type II mixed cryoglobulinemic patient are sequential phases of an antigen-driven pathological process. Int J Cancer 2000; 87: 211–216.
Pileri P, Uematsu Y, Campagnoli S, Galli G, Falugi F, Petracca R et al. Binding of hepatitis C virus to CD81. Science 1998; 282: 938–941.
Yoshida T, Hanada T, Tokuhisa T, Kosai K, Sata M, Kohara M et al. Activation of STAT3 by the hepatitis C virus core protein leads to cellular transformation. J Exp Med 2002; 196: 641–653.
Machida K, Cheng KTN, Sung VMH, Shimodaira S, Lindsay KL, Levine AM et al. Hepatitis C virus induces a mutator phenotype: enhanced mutations of immunoglobulin and protooncogenes. Proc Natl Acad Sci USA 2004; 101: 4262–4267.
Casato M, Mecucci C, Agnello V, Fiorilli M, Knight GB, Matteucci C et al. Regression of lymphoproliferative disorder after treatment for hepatitis C infection in a patient with partial trisomy 3, Bcl-2 overexpression, and type II cryoglobulinemia. Blood 2002; 99: 2259–2261.
Hermine O, Lefrere F, Bronowicki JP, Mariette X, Jondeau K, Eclache-Saudreau V et al. Regression of splenic lymphoma with villous lymphocytes after treatment of hepatitis C virus infection. N Engl J Med 2002; 347: 89–94.
Levine AM, Shimodaira S, Lai MMC . Treatment of HCV-related mantle-cell lymphoma with ribavirin and pegylated interferon alfa. N Engl J Med 2004; 349: 2078–2079.
Pitini V, Arrigo C, Righi M, Scaffidi M, Sturniolo G . Systematic screening for HCV infection should be performed in patients with splenic marginal zone lymphoma. Br J Haematol 2004; 124: 252.
Jaffe ES, Harris NL, Stein H, Vardiman JW . World Health Organization Classification of Tumours. Tumours of Haemopoietic and Lymphoid Tissue. Mature B-cell Neoplasms. Lyon: IARC Press, 2001, pp 135–138, 157–162.
Berger F, Felman P, Thieblemont C, Pradier T, Baseggio L, Bryon PA et al. Non-MALT marginal zone B-cell lymphomas: a description of clinical presentation and outcome in 124 patients. Blood 2000; 95: 1950–1956.
Macintyre EA, Belanger C, Debert C, Canioni D, Turhan AG, Azagury M et al. Detection of clonal CD34+19+ progenitors in bone marrow of BCL2-IgH-positive follicular lymphoma patients. Blood 1995; 86: 4691–4698.
Delabesse E, Burtin ML, Millien C, Madonik A, Arnulf B, Beldjord K et al. Rapid, multifluorescent TCRG Vgamma and Jgamma typing: application to T cell acute lymphoblastic leukemia and to the detection of minor clonal populations. Leukemia 2000; 14: 1143–1152.
Mazzaro C, Franzin F, Tulissi P, Pussini E, Crovatto M, Carniello GS et al. Regression of monoclonal B-cell expansion in patients affected by mixed cryoglobulinemia responsive to alpha-interferon therapy. Cancer 1996; 77: 2604–2613.
Zuckerman E, Zuckerman T, Sahar D, Streichman S, Attias D, Sabo E et al. The effect of antiviral therapy on t(14;18) translocation and immunoglobulin gene rearrangement in patients with chronic hepatitis C virus infection. Blood 2001; 97: 1555–1559.
Casato M, Agnello V, Pucillo LP, Knight GB, Leoni M, Del Vecchio S et al. Predictors of long-term response to high-dose interferon therapy in type II cryoglobulinemia associated with hepatitis C virus infection. Blood 1997; 90: 3865–3873.
Giannelli F, Moscarella S, Giannini C, Caini P, Monti M, Gragnani L et al. Effect of antiviral treatment in patients with chronic HCV infection and t(14;18) translocation. Blood 2003; 102: 1196–1201.
Luppi M, Ferrari MG, Torelli G . Hepatitis C virus genotype distribution in B-cell non-Hodgkin lymphoma. Ann Intern Med 1998; 128: 602.
Callet-Bauchu E, Gazzo S, Poncet C, Pages J, Morel D, Alliot C et al. Distinct chromosome 3 abnormalities in persistent polyclonal B-cell lymphocytosis. Genes Chromosomes Cancer 1999; 26: 221–228.
Dierlamm J, Wlodarska I, Michaux L, Stefanova M, Hinz K, Van Den Berghe H et al. Genetic abnormalities in marginal zone B-cell lymphoma. Hematol Oncol 2000; 18: 1–13.
Salcedo I, Campos-Caro A, Sampalo A, Reales E, Brieva JA . Persistent polyclonal B lymphocytosis: an expansion of cells showing IgVH gene mutations and phenotypic features of normal lymphocytes from the CD27+ marginal zone B-cell compartment. Br J Haematol 2002; 116: 662–666.
Kanchan K, Loughran Jr TP . Antigen-driven clonal T cell expansion in disorders of hematopoiesis. Leuk Res 2003; 27: 291–292.
Epling-Burnette PK, Liu JH, Catlett-Falcone R, Turkson J, Oshiro M, Kothapalli R et al. Inhibition of STAT3 signaling leads to apoptosis of leukemic large granular lymphocytes and decreased Mcl-1 expression. J Clin Invest 2001; 107: 351–362.
Metzger R, Heckl-Ostreicher B, Nerl C, Schondelmaier S, Kabelitz D . Immunological studies of gamma delta T cells in a case of large granular lymphocyte (LGL) leukemia: leukemic gamma delta+ T cells are resistant to growth stimulation in vitro but respond to interferon-alpha treatment in vivo. Leuk Res 1992; 16: 1087–1095.
Vose BM, Riccardi C, Bonnard GD, Herberman RB . Limiting dilution analysis of the frequency of human T cells and large granular lymphocytes proliferating in response to interleukin 2. II. Regulatory role of interferon on proliferative and cytotoxic precursors. J Immunol 1983; 130: 768–772.
Acknowledgements
We are grateful to Ms Phyllis Bazinet for her assistance in editing this paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kelaidi, C., Rollot, F., Park, S. et al. Response to antiviral treatment in hepatitis C virus-associated marginal zone lymphomas. Leukemia 18, 1711–1716 (2004). https://doi.org/10.1038/sj.leu.2403443
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403443
Keywords
This article is cited by
-
Development of highly aggressive mantle cell lymphoma after sofosbuvir treatment of hepatitis C
Blood Cancer Journal (2016)
-
The Role of Infectious Agents, Antibiotics, and Antiviral Therapy in the Treatment of Extranodal Marginal Zone Lymphoma and Other Low-Grade Lymphomas
Current Treatment Options in Oncology (2015)
-
Non-Hodgkin Lymphoma and Hepatitis C: Where We are and What Next?
Pathology & Oncology Research (2015)
-
microRNA levels in paraffin-embedded indolent B-cell non-Hodgkin lymphoma tissues from patients chronically infected with hepatitis B or C virus
BMC Infectious Diseases (2014)