Abstract
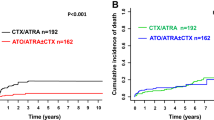
First results of a randomized trial (APL91 trial) and other randomized or non-randomized studies have shown that ATRA followed by chemotherapy significantly increased event-free survival (EFS) and survival, and decreased the incidence of relapse by comparison to chemotherapy alone in newly diagnosed APL. We present here long-term follow-up of the APL91 trial. In this trial, 101 patients had been randomized between ATRA followed by three courses of daunorubicin-AraC chemotherapy (ATRA group) and the same chemotherapy alone (chemotherapy group). Results were reanalyzed 73 months after closing of patient entry. Updated results of APL 91 trial found a Kaplan–Meier estimate of EFS and relapse rate at 4 years of 63% and 31% in the ATRA group, as compared to 17% and 78% in the chemotherapy group (P = 10−4 and relative risk 2.95, P = 10−4 and relative risk 3.68, respectively). Kaplan–Meier survival at 4 years was 76% in the ATRA group and 49% in the chemotherapy group (P = 0.026, relative risk 2.7). In the chemotherapy group, seven of the 27 relapses occurred after 18 months, but no relapse was seen after 43 months. In the ATRA group, four of the 17 relapses occurred after 18 months, including two late relapses (at 58 and 74 months). In the chemotherapy group, 23 of the 25 patients who relapsed achieved a second CR with ATRA, and the Kaplan–Meier estimate of second relapse was 40% at 30 months. In the ATRA group, the 10 patients who relapsed and were retreated with ATRA achieved a second CR. In conclusion, long-term results of APL91 trial confirm the superiority of the combination of ATRA and chemotherapy over chemotherapy alone in newly diagnosed APL, and that ATRA should be incorporated in the front-line treatment of APL.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton D, Gralnick M, Sultan C . Proposals for the classification of the acute leukemias Br J Haematol 1976 33: 451–461
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton D, Gralnick M, Sultan C . A variant form of hypergranular promyelocytic leukemia (M3) Ann Intern Med 1980 92: 280–288
Larson RA, Kondo K, Vardiman JW, Butler AR, Golomb HM, Rowley JD . Evidence for a 15;17 translocation in every patient with acute promyelocytic leukemia Am J Med 1984 76: 827–835
De The H, Lavau C, Marchio A, Chomienne C, Degos L, Dejean A . The PML-RAR alpha fusion mRNA generated by the t(15;17) translocation in acute promyelocytic leukemia encodes a functionally altered RAR Cell 1991 66: 675–686
Kakizuka A, Miller WH, Umesono K, Warrell R, Frankel S, Dmitrovsky E, Evans R . Chromosomal translocation t(15;17) in human acute promyelocytic leukemia fuses RAR alpha with a novel putative transcription factor, PML Cell 1991 66: 663–674
Tallman MS, Kwaan HC . Reassessing the hemostatic disorder associated with acute promyelocytic leukemia Blood 1992 79: 543–553
Dombret H, Sutton L, Duarte M, Daniel MT, Leblond V, Castaigne S, Degos L . Combined therapy with all-trans retinoic acid and high-dose chemotherapy in patients with hyperleukocytic acute promyelocytic leukemia and severe visceral hemorrhage Leukemia 1992 6: 1237–1242
Goldberg MA, Ginsburg D, Mayer RJ, Stone RM, Maguire M, Rosenthal DS, Antin JH . Is heparin administration necessary during induction chemotherapy for patients with acute promyelocytic leukemia? Blood 1987 69: 187–191
Cunningham I, Gee T, Reich L, Kempin SJ, Naval AN, Clarkson BD . Acute promyelocytic leukemia: treatment results during a decade at Memorial Hospital Blood 1989 73: 1116–1122
Rodighiero F, Avvisati G, Castaman G, Mandelli F . Early deaths and anti-hemorrhagic treatments in acute promyelocytic leukemia. A GIMEMA retrospective study in 268 consecutive patients Blood 1990 11: 2112–2117
Fenaux P, Pollet JP, Vandenbossche L, Dupriez B, Jouet JP, Bauters F . Treatment of acute promyelocytic leukemia: a report on 70 cases Leuk Lymphoma 1991 4: 249–256
Head D, Kopecky KJ, Willman CL, Appelbaum F, Tao RF, Xia XM, Li C, Wang W, Zu WY, Yao XZ, Ling BJ . Aggressive daunomycin (DNR) therapy improves survival in acute promyelocytic leukemia (APL), a Southwest Oncology Group (SWOG trial) Leukemia 1994 8: (Suppl.2) S38–S41
Huang M, Yu-Chen Y, Shu-Rom GC, Lu MX, Zhoa L, Gu LJ, Wang ZY . Use of all-trans retinoic acid in the treatment of acute promyelocytic leukemia Blood 1988 72: 567–572
Chomienne C, Ballerini P, Balitrand N, Daniel MT, Fenaux P, Castaigne S, Degos L . All-trans retinoic acid in promyelocytic leukemias. II. In vitro studies structure function relationship Blood 1990 76: 1710–1717
Castaigne S, Chomienne C, Daniel MT, Berger R, Fenaux P, Degos L . All-trans retinoic acid as a differentiating therapy for acute promyelocytic leukemias. I. Clinical results Blood 1990 76: 1704–1709
Warrel RP, Frankel SR, Miller W, Itri L, Andreeff M, Jabukowski A, Gabrilove J, Gordon MS, Dmitrovsky E . Differentiation therapy of acute promyelocytic leukemia with tretinoin (all-trans retinoic acid) New Engl J Med 1991 324: 1385–1394
Chen ZX, Xue YQ, Zhang R, Tao RF, Xia XM, Li C, Wang W, Zu WY, Yao XZ, Ling BJ . A clinical and experimental study on all-trans retinoic acid treated acute promyelocytic leukemia patients Blood 1991 78: 1413–1419
Degos L, Chomienne C, Daniel MT, Berger R, Dombret H, Fenaux P, Castaigne S . Treatment of first relapse in acute promyelocytic leukaemia with all-trans retinoic acid Lancet 1990 2: 1440–1441
Fenaux P, Le Deley MC, Castaigne S, Archimbaud E, Chomienne C, Link H, Guerci A, Duarte M, Daniel MT, Bowen D, Huebner G, Bauters F, Fegueux N, Fey M, Sanz M, Lowenberg B, Maloisel F, Auzanneau G, Sadoun A, Gardin C, Bastion Y, Ganser A, Jacky E, Dombret H, Chastang C, Degos L . Effect of all-trans retinoic acid in newly diagnosed acute promyelocytic leukemia. Results of a multicenter randomized trial Blood 1993 82: 3241–3249
Tallman MS, Andersen JW, Schiffer CA, Appelbaum FR, Feusner JH, Ogden A, Shepherd L, Willman C, Bloomfield CD, Rowe JM, Wiernik PH . All-trans retinoic acid in acute promyelocytic leukemia New Engl J Med 1997 337: 1021–1028
Kanamaru A, Takemoto Y, Tanimoto M, Murakami H, Asou N, Kobayashi T, Kuriyama K, Ohmoto E, Sakamaki H, Tsubaki K . All-trans retinoic acid for the treatment of newly diagnosed acute promyelocytic leukemia Blood 1995 85: 1202–1206
Avvisatti G, Baccarani M, Ferrara F, Lazzarino M, Resegotti L, Mandelli F . AIDA protocol (all-trans retinoic acid+idarubicin) in the treatment of newly diagnosed acute promyelocytic leukemia (APL): a pilot study of the Italian cooperative group GIMEMA Blood 1994 84: (Suppl.1) 380a (Abstr.)
Fenaux P, Castaigne S, Dombret H, Archimbaud E, Duarte M, Morel P, Lamy T, Tilly H, Guerci A, Maloisel F, Bordessoule D, Sadoun A, Tiberghien P, Fegueux N, Daniel MT, Chomienne C, Degos L . All-trans retinoic acid followed by intensive chemotherapy gives a high complete remission rate and may prolong remissions in newly diagnosed acute promyelocytic leukemia. A pilot study on 26 cases Blood 1992 80: 2176–2181
Warrel RP, Maslak P, Eardley A, Heller G, Miller WH, Frankel SR . Treatment of acute promyelocytic leukemia with all-trans retinoic acid: an update of the New York experience Leukemia 1994 8: 926–933
Kaplan E, Meier P . Nonparametric estimation from incomplete observations J Am Stat Assoc 1958 53: 457–472
Cox DR . Regression models and life-tables (with discussion) JR Stat Soc B 1972 34: 487–499
Peto R, Peto J . Asymptotically efficient rank invariant test procedures (with discussion) JR Stat Soc A 1972 135: 185–194
Fenaux P, Chastang C, Chomienne C, Degos L . Tretinoin with chemotherapy in newly diagnosed acute promylocytic leukaemia Lancet 1994 343: 1033–1034
Burnett AK, Grimwade D, Solomon E, Wheatley K, Goldstone AH . Presenting white blood cell count and kinetics of molecular remission predict prognosis in acute promyelocytic leukemia treated with all-trans retinoic acid: result of the randomized MRC trial Blood 1999 93: 4131–4143
Fenaux P, Chastang C, Chevret S, Sanz M, Dombret H, Archimbaud E, Fey M, Rayon C, Huguet F, Sotto JJ, Gardin C, Makhoul PC, Travade P, Solary E, Fegueux N, Bordessoule D, Miguel JS, Link H, Desablens B, Stamatoullas A, Deconinck E, Maloisel F, Castaigne S, Preudhomme C, Degos L . A randomized comparison of all-trans retinoic acid (ATRA) followed by chemotherapy and ATRA plus chemotherapy and the role of maintenance therapy in newly diagnosed acute promyelocytic leukemia. The European APL Group Blood 1999 94: 1192–1200
Vahdat L, Maslak P, Miller WH, Eardley A, Heller, Scheinberg DA, Warrell RP . Early mortality and the retinoic acid syndrome in acute promyelocytic leukemia: impact of leukocytosis, low-dose chemotherapy, PML/RAR isoform, and CD13 expression in patients treated with all-trans retinoic acid Blood 1994 84: 3843–3849
Fukutani H, Naoe T, Ohno R, Yoshida H, Kiyoi H, Miyawaki S, Morishita H, Sano F, Kamibayashi H, Matsue K . Prognostic significance of the RT-PCR assay of PML-RARα transcripts in acute promyelocytic leukemia. The Leukemia Study Group of the Ministry of Health and Welfare (Kouseisho) Leukemia 1995 4: 588–593
Miller WH, Levine K, Deblasio A, Frankel SR, Dmitrovsky E, Warrel RP . Detection of minimal residual disease in acute promyelocytic leukemia by a reverse transcription polymerase chain reaction assay for PML-RAR fusion mRNA Blood 1993 82: 1689–1694
Ikeda K, Sasaki K, Tasaka T, Nagai M, Kawanishi K, Takahara J, Irino S . Reverse transcription polymerase chain reaction for PML-RAR fusion transcripts in acute promyelocytic leukemia and its application to minimal residual detection Leukemia 1993 7: 544–548
Diverio D, Pandolfi PP, Rossi V, Biondi A, Pelicci PG, Lo Coco F . Monitoring of treatment outcome in acute promylelocytic leukemia by RT-PCR Leukemia 1994 8: 1105–1107
Muindi J, Frankel SR, Miller WH, Jakubowski A, Scheinberg DA, Young CW, Dimitrovsky E . Continuous treatment with all-trans RA progressively decreases plasma drug concentrations: implications for relapse and resistance in acute promyelocytic leukemia Blood 1992 79: 299–303
Delva L, Cornic M, Balitrand N, Delva L, Cornic M, Balitrand N, Guidez F, Miclea JM, Delmer A, Teillet F, Fenaux P, Castaigne S, Degos L, Chomienne C . Resistance to all-trans retinoic acid (ATRA) therapy in relapsing acute promyelocytic leukemia: study of in vitro ATRA sensitivity and cellular retinoic acid binding protein levels in leukemic cells Blood 1993 82: 2175–2181
Shen ZX, Chen GQ, Ni JH, Li XS, Xiong SM, Qiu QY, Zhu J, Tang W, Sun GL, Yang KQ, Chen Y, Zhou L, Fang ZW, Wang YT, Ma J, Zhang P, Zhang TD, Chen SJ, Chen Z, Wang ZY . Use of arsenic trioxide (As2O3) in the treatment of acute promyelocytic leukemia (APL): II. Clinical efficacy and pharmacokinetics in relapsed patients Blood 1997 89: 3354–3360
Soignet SL, Maslak P, Wang ZG, Jhanwar S, Calleja E, Dardashti LJ, Corso D, Deblasio A, Gabrilove J, Scheinberg DA, Pandolfi PP, Warrell RP Jr . Complete remission after treatment of acute promyelocytic leukemia with arsenic trioxide New Engl J Med 1998 339: 1341–1348
Sanz M, Martin G, Rayon C, Esteve J, Gonzalez M, Diaz-Mediavilla J, Bolufer P, Barragan E, Terol MJ, Gonzalez JD, Colomer D, Chillon C, Rivas C, Gomez T, Ribera JM, Borstein R, Roman J, Calasanz MJ, Arias J, Alvarez C, Ramos F, Deben G . A modified AIDA protocol with anthracyclin based consolidation results in high antileukemic efficacy and reduced toxicity in newly diagnosed PML-RAR α-positive acute promyelocytic leukemia Blood 1999 94: 3015–3021
Lo Coco F, Diverio D, Avvisati G, Petti MC, Meloni G, Pogliani EM, Biondi A, Rossi G, Carlo-Stella C, Selleri C, Martino B, Specchia G, Mandelli F . Therapy of molecular relapse in acute promyelocytic leukemia Blood 1999 94: 2225–2229
Acknowledgements
This work was supported by the Programme Hospitalier de Recherche Clinique, Centre Hospitalier Universitaire (CHU) of Lille, and the Association pour la Recherche contre le Cancer (ARC).
Author information
Authors and Affiliations
Consortia
Appendix
Appendix
Dr P Fenaux and Dr L Degos served as cochairmen, and Dr C Chastang and S Chevret-Chastang (Department of Biostatistics, Hopital St Louis, Paris) as biostatisticians.
The following clinical departments participated in the APL93 trial.
French APL group
S Castaigne, H Dombret (Paris), R Zittoun (Paris), E Archimbaud (Lyon), P Travade (Clermont Ferrand), C Gardin (Clichy), A Guerci (Nancy), P Fenaux (Lille), AM Stoppa (Marseille), F Dreyfus (Paris), F Stamatoulas (Rouen), F Rigal-Huguet (Toulouse), H Guy (Dijon), JJ Sotto (Grenoble), F Maloisel (Strasbourg), J Reiffers (Pessac), A Gardembas (Angers), D Bordessoule (Limoges), N Fegueux (Montpellier), A Buzyn (Paris), T Lamy (Rennes), Ph Brault (Villejuif), E Deconinck (Besançon), E Guyotat (St Etienne), M Martin (Annecy), E Cony-Makhoul (Bordeaux), JP Abgrall (Brest), O Reman (Caen), B Desablens (Amiens), JL Harousseau (Nantes), Y Bastion (Lyon), JP Pollet (Valenciennes), J Pulik (Argenteuil), M Lepeu (Avignon), M Renoux (Bayonne), P Morel (Lens), P Henon (Mulhouse), N Gratecos (Nice), P Colombat (Tours), D Machover (Villejuif), A Dor (Antibes), P Casassus (Bobigny), J Donadio (Castelnou), B Salles (Chalon), B Legros (Clermont Ferrand), P Audhuy (Colmar), A Dutel (Compiègne), N Philippe (Lyon), B Benothman (Meaux), C Christian (Metz), C Margueritte (Montpellier), F Witz (Nancy), A Pesce (Nice), A Baruchel (Paris), L Sutton (Paris), C Quetin (Pointe à Pitre), B Pignon (Reims), E Vilmer (Paris), E Bourquard (St Brieuc), JP Marolleau (Paris), P Robert (Toulouse), B Despax (Toulouse), G Nedellec, P Auzanneau (Paris), M Janvier (St Cloud).
Spanish AML group
O Rayon (Oviedo), M Sanz (Valencia), J San Miguel (Salamanca), J Montagud (Valencia), E Condé (Santander), P Javier de la Serna (Madrid), G Martin (Valencia), M Perez Encinas (Santiago), JP Torres Carrete (Juan Canalejo), J Zuazu (Barcelone), J Odriozola (Madrid), E Gomez-Sanz (Madrid), L Palomera (Zaragoza), L Villegas (Almeria), A Deben (Juan Canalejo), P Besalduch (Palma de Mallorca).
Cooperative AML study group, Germany
H Link (Hannover), A Ganser (Frankfurt), H Wandt (Nurnberg), A Breitenbach (Stuttgart), B Brennscheidt (Freiburg), D Herrmann (Ulm), H Soucek (Dresden), H Strobel (Erlangen)
SAKK Swiss AML group
K Geiser (Berne), M Fey (Berne), T Egger (Berne), E Jacky
Belgian groups
JL Michaux (Bruxelles), A Bosly (Yvoir), E Meeus (Anvers), A Boulet (Mons), Ph Mineur (Gilly)
Dutch groups
P Daenen (Groningen), P Muus (Nijmegen)
British group
JA Whittaker (Cardiff)
Rights and permissions
About this article
Cite this article
Fenaux, P., Chevret, S., Guerci, A. et al. Long-term follow-up confirms the benefit of all-trans retinoic acid in acute promyelocytic leukemia. Leukemia 14, 1371–1377 (2000). https://doi.org/10.1038/sj.leu.2401859
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2401859
Keywords
This article is cited by
-
Analysis of risk factors for early death in patients with acute promyelocytic leukaemia treated with arsenic trioxide
Annals of Hematology (2022)
-
Retrospective analysis of data from 73 patients with childhood acute promyelocytic leukaemia receiving modified chemotherapy: a single-centre study
Journal of Cancer Research and Clinical Oncology (2021)
-
Arsenic trioxide-based therapy of relapsed acute promyelocytic leukemia: registry results from the European LeukemiaNet
Leukemia (2015)
-
Overview of Therapy and Strategies for Optimizing Outcomes in De Novo Pediatric Acute Myeloid Leukemia
Pediatric Drugs (2014)
-
Stemness and inducing differentiation of small cell lung cancer NCI-H446 cells
Cell Death & Disease (2013)