Abstract
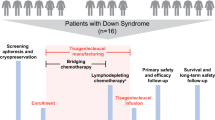
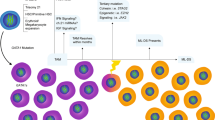
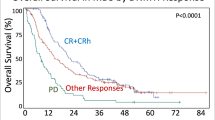
In recent pediatric collaborative studies of acute myeloid leukemia (AML), patients with Down's syndrome (DS) have better outcome than other patients when they were treated according to their intensive AML protocols. This may be attributed to enhanced sensitivity of DS AML cells to selected chemotherapeutic agents. We evaluated a less intensive chemotherapeutic regimen which was specifically designed for children with AML-DS. Remission induction chemotherapy consisted of daunorubicin (25 mg/m2/day for 2 days), cytosine arabinoside (100 mg/m2/day for 7 days), and etoposide (150 mg/m2/day for 3 days). Patients received one to seven courses of consolidation therapy of the same regimen. Thirty-three patients were enrolled on the study and their clinical, hematologic and immunophenotypic features were analyzed. Of the 33 patients, all were younger than 4 years and diagnosed as having acute megakaryoblastic leukemia or myelodysplastic syndrome. All patients achieved a complete remission and estimated 8 year event-free survival rate was 80 ± 7%. Three patients relapsed and two died due to cardiac toxicity and one due to septic shock. The results of our study showed that patients with AML-DS constitute a unique biologic subgroup and should be treated according to a less intensive protocol designed for AML-DS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Robinson LL . Down syndrome and leukemia Leukemia 1992 6: 5–7
Avet-Loiseau H, Mechinaud F, Harousseau J-L . Clonal hematologic disorders in Down syndrome J Pediatr Hematol Oncol 1995 17: 19–24
Kojima S, Matsuyama T, Sato T, Horibe K, Konishi S, Tsuchida M, Hayash Y, Kigasawa H, Akiyama Y, Okamura J, Nakahata T, Bessho F, Eguchi M, Nakazawa S, Ueda R . Down's syndrome and acute leukemia in children: an analysis of phenotype by use of monoclonal antibodies and electron microscopic platelet peroxidase reaction Blood 1990 76: 2348–2353
Ravindranath Y, Abella E, Krischer JP, Wiley J, Inoue S, Harris M, Chauvenet A, Alvarado CS, Dubowy R, Ritchey AK, Land V, Steuber CP, Weinstein H . Acute myeloid luekemia (AML) in Down's syndrome is highly responsive to chemotherapy: experience on Pediatric Oncology Group AML Study 8498 Blood 1992 80: 2210–2214
Lie SO, Jonmundsson G, Mellander L, Siimes MA, Yssing M, Gustafsson G . A population-based study of 272 children with acute myeloid leukaemia treated on two consecutive protocols with different intensity: best outcome in girls, infants, and children with Down's syndrome Br J Haematol 1996 94: 82–88
Creutzig U, Ritter J, Vormoor J, Ludwig WD, Niemeyer C, Reinisch I, Stollmann-Gibbels B, Zimmermann M, Harbott J . Myelodysplasia and acute myelogenous leukemia in Down's syndrome. A report of 40 children of the AML-BFM study group Leukemia 1996 10: 1677–1686
Stevens RF, Hann IM, Wheatley K, Gray RG . Marked improvements in outcome with chemotherapy alone in paediatric acute myeloid leukaemia: results of the United Kingdom Medical Research Council's 10th AML trial Br J Haematol 1998 101: 130–140
Lange BJ, Kobrinsky N, Barnard DR, Arthur DC, Buckley JD, Howells WB, Gold S, Sanders J, Neudorf S, Smith FO, Woods WG . Distinctive demography, biology, and outcome of acute myeloid leukemia and myelodysplastic syndrome in children with Down syndrome: Children's Cancer Group Studies 2861 and 2891 Blood 1998 91: 608–615
Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DAG, Gralnick HR, Sultan C . The French–American–British (FAB) Co-operative Group: Proposals for the classification of the myelodysplastic syndromes Br J Haematol 1982 51: 189–199
Eguchi M, Sakakibara H, Suda J, Ozawa T, Hayashi Y, Sato T, Kojima S, Furukawa T . Ultrastructural and ultracytochemical differences between transient myeloproliferative disorder and megakaryoblastic leukaemia in Down's syndrome Br J Haematol 1989 73: 315–332
Kaplan EL, Meier P . Non-parametric estimation from incomplete observations J Am Stat Assoc 1958 53: 457–481
Cheson BD, Cassileth PA, Head DR, Schiffer CA, Bennett JM, Bloomfield CD, Brunning R, Gale RP, Grever MR, Keating MJ . Report of the National Cancer Institute-sponsored workshop on definitions of diagnosis and response in acute myeloid leukemia J Clin Oncol 1990 8: 813–819
Yumura-Yagi K, Hara J, Kurahashi H, Nishiura T, Kaneyama Y, Osugi Y, Sakata N, Inoue M, Tawa A, Okada S, Kawa-Ha K . Mixed phenotype of blasts in acute megakaryocytic leukaemia and transient abnormal myelopoiesis in Down's syndrome Br J Haematol 1992 81: 520–525
Slordahl SH, Smeland EB, Holte H, Gronn M, Lie SO, Seip M . Leukemic blasts with markers of four cell lineages in Down's syndrome (‘Megakaryoblastic Leukemia’) Med Pediatr Oncol 1993 21: 254–258
Okumura N, Tsuji K, Nakahata T . Changes in cell surface antigen expressions during proliferation and differentiation of human erythroid progenitors Blood 1992 80: 642–650
Ito E, Kasai M, Hayashi Y, Toki T, Arai K, Yokoyama S, Kato K, Tachibana N, Yamamoto M, Yokoyama M . Expression of erythroid-specific genes in acute megakaryoblastic leukaemia and transient myeloproliferative disorder in Down's syndrome Br J Haematol 1995 90: 607–614
Kita K, Miwa H, Nakase K, Kawakami K, Kobayashi T, Shirakawa S, Tanaka I, Ohta C, Tsutani H, Oguma S, Kyo T, Dohy H, Kamada N, Nasu K, Uchino H . Clinical importance of CD7 expression in acute myelocytic leukemia Blood 1993 81: 2399–2405
Creutzig U, Harbott J, Sperling C, Ritter J, Zimmermann M, Loffler H, Riehm H, Schellong G, Ludwig W-D . Clinical significance of surface antigen expression in children with acute myeloid leukemia: results of Study AML-BFM-87 Blood 1995 86: 3097–3108
McDonald JP, Sullivan PS . Megakaryocytic and erythrocytic cell lines share a common precursor cell Exp Hematol 1993 21: 1316–1328
Skipper HE, Schable JM, Wilcox WS . Experimental evaluation of potential anticancer agents: scheduling of arabinosylcytosine to take advantage of its S-phase specificity against leukemia cells Cancer Chemother Rep 1967 51: 125–165
Taub JW, Matherly LH, Stout ML, Buck SA, Gurney JG, Ravindranath Y . Enhanced metabolism of 1-β-D-arabinofuranosylcytosine in Down syndrome cells: a contributing factor to the superior event free survival of Down syndrome children with acute myeloid leukemia Blood 1996 87: 3395–3403
Fong C, Brodeur GM . Down's syndrome and leukemia: epidemiology, genetics, cytogenetics and mechanisms of leukemogenesis Cancer Genet Cytogenet 1987 28: 55–76
Gilladoga AC, Manuel C, Tan CTC, Wollner N, Sternberg SS, Murphy ML . The cardiotoxicity of adriamycin and daunomycin in children Cancer 1976 37: 1070–1078
Lewis AB, Pilkington R, Takahashi M, Siegel SE . Echocardiographic assessment of anthracycline cardiotoxicity in children Med Pediatr Oncol 1978 5: 167–175
Lipshultz SE, Colan SD, Gelber RD, Perez-Atayde AR, Sallan SE, Sanders SP . Late cardiac effects of doxorubicin therapy for acute lymphoblastic leukemia in childhood New Engl J Med 1991 324: 808–815
Hayashi Y, Eguchi M, Sugita K, Nakazawa S, Sato T, Kojima S, Bessho F, Konishi S, Inaba T, Hanada R, Yamamoto K . Cytogenetic findings and clinical features in acute leukemia and transient myeloproliferative disorder in Down's syndrome Blood 1988 72: 15–23
Haas OA, Gadner H . Pathogenesis, biology, and management of myelodysplastic syndromes in children Semin Hematol 1996 33: 225–235
Woods WG, Nesbit ME, Buekley J, Lampkin BC, McCreadie S, Kim TH, Piomelli S, Kersy JH, Feig S, Bernstein I, Hammond D, the Children's Cancer Study Group . Correlation of chromosome abnormalities with patient characteristics, histologic subtype, and induction success in children with acute nonlymphocytic leukemia J Clin Oncol 1985 3: 3–11
Kalwinsky DK, Raimondi SC, Schell MJ, Mirro JJ, Santana VM, Behm F, Dahl GV, Williams D . Prognostic importance of cytogenetic subgroups in de novo pediatric acute nonlymphocytic leukemia J Clin Oncol 1990 8: 75–83
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kojima, S., Sako, M., Kato, K. et al. An effective chemotherapeutic regimen for acute myeloid leukemia and myelodysplastic syndrome in children with Down's syndrome. Leukemia 14, 786–791 (2000). https://doi.org/10.1038/sj.leu.2401754
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2401754
Keywords
This article is cited by
-
Clinical and biological aspects of myeloid leukemia in Down syndrome
Leukemia (2021)
-
Anthracycline-Related Cardiotoxicity in Patients with Acute Myeloid Leukemia and Down Syndrome: A Literature Review
Cardiovascular Toxicology (2016)
-
Mosaic Down syndrome-associated acute myeloid leukemia does not require high-dose cytarabine treatment for induction and consolidation therapy
International Journal of Hematology (2010)
-
High Erk-1 activation and Gadd45a expression as prognostic markers in high risk pediatric haemolymphoproliferative diseases
Journal of Experimental & Clinical Cancer Research (2009)
-
The role of the proto-oncogene ETS2 in acute megakaryocytic leukemia biology and therapy
Leukemia (2008)