Abstract
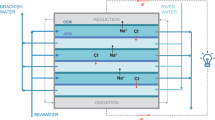
WHEN a hypertonic solution is separated from a hypotonic one by a semipermeable membrane, solvent will flow from the hypotonic to the hypertonic system until equilibrium is established. Sea water is essentially a 0.6 N solution of polar solutes in water. A membrane which would, under certain conditions, reject sodium and larger ions and allow migration of water free of solutes can be obtained1. Cellulose acetate, for example, fulfils the condition of rejection of sodium chloride2. A semi-permeable membrane system for concentrating beverages was conceived some 30 years ago3, but practical development has awaited the advent of appropriate membranes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Popper, K., Stanley, W. L., Nury, F. S., and Kohler, G. O. (in preparation).
Reid, C. E., and Breton, D. J., J. App. Polymer Sci., 1 (2), 133 (1959).
Wickenden, L., U.S. Patent No. 2,116,920, May 10, 1938.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
POPPER, K., CAMIRAND, W., STANLEY, W. et al. Osmotic Emergency Purifier of Sea Water. Nature 211, 297–298 (1966). https://doi.org/10.1038/211297a0
Issue Date:
DOI: https://doi.org/10.1038/211297a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.