Abstract
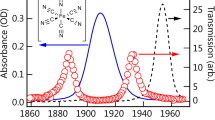
NITRENES, molecular fragments with an electron sextet on nitrogen, are known to be the primary products of the decomposition of organic azides1–3. Their existence as separate intermediates and their triplet character were recently demonstrated by electron spin resonance spectroscopy4. We have investigated the photolysis of several aromatic azides and diazides in organic glasses at 77° K in an attempt to trap the nitrenes presumably formed. Three types of behaviour were observed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Boyer, J. H., and Canter, F. C., Chem. Revs., 54, 1 (1954).
Smolinsky, G., J. Org. Chem., 26, 4108 (1961).
Horner, L., and Christman, A., Angew. Chemie. (Intern. Ed.), 2, 599 (1963).
Smolinsky, G., Wasserman, E., and Yager, W. A., J. Amer. Chem. Soc., 84, 3220 (1962).
Norman, I., and Porter, G., Proc. Roy. Soc., A, 230, 399 (1955).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
REISER, A., FRAZER, V. Ultra-violet Absorption Spectra of Aromatic Nitrenes and Dinitrenes. Nature 208, 682–683 (1965). https://doi.org/10.1038/208682a0
Issue Date:
DOI: https://doi.org/10.1038/208682a0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.