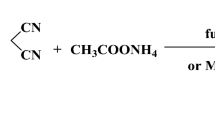Abstract
IN an earlier paper on this topic1 we proposed a four-parameter equation (1), which expresses the quantitative relationship between the structure of p-disubstituted derivatives of benzene and the magnitude of their biological activity, for example intravenous LD50. This report presents our results on a group of compounds of the type  NO2, NH2. The series includes all possible combinations of groups X and Y, with the exception of three substances which were not soluble enough even in 20 per cent aqueous polyvinylpyrrolidone solution to make LD50 measurements possible. The experimental results fitted equation
NO2, NH2. The series includes all possible combinations of groups X and Y, with the exception of three substances which were not soluble enough even in 20 per cent aqueous polyvinylpyrrolidone solution to make LD50 measurements possible. The experimental results fitted equation
Similar content being viewed by others
Article PDF
References
Boček, K., Kopecký, J., Krivucová, M., and Vlachová, D., Experientia, 20, 667 (1964).
Hammett, L. P., Physical Organic Chemistry (McGraw-Hill, New York, 1940).
Taft, jun., R. W., J. Amer. Chem. Soc., 75, 4231 (1953).
Zahradník, R., Experientia, 18, 534 (1962).
Zahradník, R., Arch. Int. Pharmacodyn., 135, 311 (1962).
Hansch, C., Maloney, P. P., Fujita, T., and Muir, R. M., Nature, 194, 178 (1962).
Hansch, C., Muir, R. M., Fujita, T., Maloney, P. P., Geiger, F., and Streich, M., J. Amer. Chem. Soc., 85, 2817 (1963).
Hansch, C., and Fujita, T., J. Amer. Chem. Soc., 86, 1616 (1964).
Bruice, T. C., Kharasch, N., and Winzler, R. J., Arch. Biochem. Biophys., 62, 305 (1956).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
KOPECKÝ, J., BOČEK, K. & VLACHOVÁ, D. Chemical Structure and Biological Activity on m-and p-Disubstituted Derivatives of Benzene. Nature 207, 981 (1965). https://doi.org/10.1038/207981a0
Issue Date:
DOI: https://doi.org/10.1038/207981a0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.