Abstract
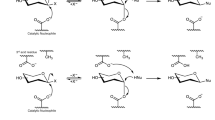
INVESTIGATIONS into the specificity of β-xylosidase required the synthesis of a number of substituted phenyl-β-D-xylosides. A very important method is the Helferich reaction in which the appropriate phenol and the fully acetylated monosaccharide are heated in the presence of an acidic catalyst. To favour the formation of xyloside-acetates with the β-anomeric configuration, p-toluene-sulphonic acid and β-tetra-O-acetyl-xylose were used1.
Similar content being viewed by others
Article PDF
References
Montgomery, E. M., Richtmeyer, N. K., and Hudson, C. S., J. Amer. Chem. Soc., 64, 690 (1942).
Vogel, A. I., A Textbook of Practical Organic Chemistry, third ed., 452 (London, Longmans, Green and Co., 1957).
Jermyn, M. A., Austral. J. Chem., 8, 403 (1955).
Loontiens, F. G., and De Bruyne, C. K., Naturwiss., 51, 359 (1964).
Thompson, A., and Wolfrom, M. L., in Methods of Carbohydrate Chemistry, 2, 215, edit. by Whistler, R. L., and Wolfrom, M. L. (New York, Academic Press, Inc., 1963).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DE BRUYNE, C., VERSELE, H. & CLAEYSSENS, M. Synthesis of Substituted Phenyl-β-D-xylo-pyranosides. Nature 205, 900 (1965). https://doi.org/10.1038/205900a0
Issue Date:
DOI: https://doi.org/10.1038/205900a0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.