Abstract
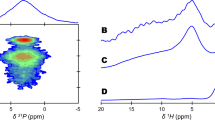
USING a method of centrifugation in liquids progressively increasing in specific gravity1, it is possible to separate, from a sample of bone powder, fractions at various degrees of mineralization2. These fractions, which can be recovered quantitatively, can then be analysed by appropriate chemical and biophysical techniques. We used infra-red spectroscopy for the examination of the carbonate present in bone, comparatively for a low specific gravity fraction (1.7 g/c.c.) and a pool of the high specific gravity fractions (1.8–2.3 g/c.c.) derived from diaphyseal bone samples of normal rats. It must be pointed out that less than 0.5 per cent of the samples belonged to the 1.7 gravity fraction. We used a Perkin–Elmer 221 infra-red spectrophotometer equipped with a sodium chloride prism. Potassium bromide pellets were prepared with various concentrations of whole unashed diaphyseal bone after separation.
Similar content being viewed by others
Article PDF
References
Herman, H., and Richelle, L., Bull. Soc. Chim. biol., 43, 273 (1961).
Richelle, L., Clin. Orthop., 33, 211 (1964).
Cabannes, J., Rev. Sci., 80, 400 (1942).
Emerson, W. H., and Fisher, E. E., Arch. Oral Biol., 7, 671 (1962).
Elliott, J. C., Arch. Oral Biol., ORCA Suppl., 277 (1963).
Herman, H., and Dallemagne, M. J., Bull. Soc. Chim. biol., 46, 373 (1964).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
RICHELLE, L., QUINAUX, N. Presence of Carbonate in a Low-density Fraction of Diaphyseal Bone. Nature 203, 84 (1964). https://doi.org/10.1038/203084a0
Published:
Issue Date:
DOI: https://doi.org/10.1038/203084a0
This article is cited by
-
Bone Carbonate and the Ca to P Molar Ratio
Nature (1968)
-
Synthetic hydroxyapatite crystals
Calcified Tissue Research (1967)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.