Abstract
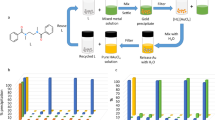
THE radioisotope zinc-65 is often used as a tracer element for investigations of transport phenomena in metals. It is conveniently introduced by electroplating the zinc on to a cathode made of one of the components of the material to be investigated. Unfortunately, no simple and flexible plating procedure has yet been used for this work. Shirn, Wajda and Huntington1 developed the procedure used for their own self-diffusion studies, and Jaumot and Smith2 used the same procedure. However, this requires a rotating cathode, a zinc anode, and a relatively high concentration of zinc-65. Also, the plating efficiency is low—about 6 per cent.
Similar content being viewed by others
Article PDF
References
Shirn, G. A., Wajda, E. S., and Huntington, H. B., Acta Met., 1, 513 (1953).
Jaumot, jun., F. E., and Smith, R. L., Trans. Amer. Inst. Min., Metall., Petrol. Eng., 206, 164 (1956).
Shteinberg, B., and Bogakovskii, N. A., Vestn. Mashinostr., 41, No. 8 41 (1962); Chem. Abstr. 2136d (1963).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
RIGNEY, D. Plating Radioactive Zinc-65. Nature 201, 1021 (1964). https://doi.org/10.1038/2011021a0
Issue Date:
DOI: https://doi.org/10.1038/2011021a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.