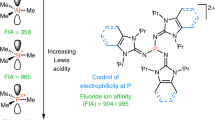
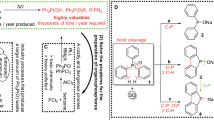
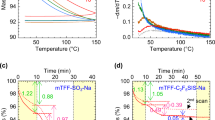
Abstract
IN a recent series of papers1, Peppard et al. have examined alkyl and aryl phosphoric and phosphonic acids and their esters. As a result of cryoscopic and infra-red measurements, they have concluded that, except in acetic acid solution, organophosphorus acids are dimeric, or polymeric, depending on the solvent, and that there is no evidence of the monomer in dilute solutions (M/1,000) in carbon tetrachloride. They conclude that the hydrogen bonding in these acids is stronger than that in carboxylic acids. These conclusions are in agreement with those reached by earlier workers2–5, who all report the bonding to be unaffected by dilution in non-polar solvents. In view of these findings, it has been suggested3,6 that the hydrogen bonding may be intramolecular.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Peppard, D. F., Ferraro, J. R., and Mason, G. W., J. Inorg. Nucl. Chem., 4, 371 (1957).; 7, 231 (1958); 12, 60 (1959); 16, 246 (1960).
Bellamy, L. J., Infrared Spectra of Complex Molecules, second ed. (Methuen, London, 1959).
Maarsen, J. W., thesis, University of Amsterdam (1956).
Jones, R. N., and Sandorfy, Camille, “The Application of Infrared and Raman Spectrometry to the Elucidation of Molecular Structure”; Part 4 of Weissberger's Chemical Applications of Spectroscopy (Intersci. Pub., London, 1956).
Freedman, L. W., and Doak, G. O., Chem. Rev., 57 (3), 479 (1957).
Thomas, L. C., Chem. and Indust., 198 (1957).
Corbridge, D. E. C., J. App. Chem., 6, 456 (1956).
Ryskin, Ya. I., and Stavitskaya, G. P., Optics and Spectroscopy, 7, 488 (1959).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
THOMAS, L., CHITTENDEN, R. & HARTLEY, H. Hydrogen Bonding in Alkyl Phosphonic and Alkyl Phosphonothionic Acids. Nature 192, 1283–1284 (1961). https://doi.org/10.1038/1921283a0
Issue Date:
DOI: https://doi.org/10.1038/1921283a0
This article is cited by
-
Synthesis, X-ray diffraction, thermal behavior and physicochemical studies of phase transitions before the decomposition in the new phosphate tellurate protonic conductor compound
Research on Chemical Intermediates (2021)
-
IR Spectra and the structure of di-2-ethylhexylphosphoric acid and its H-complexes with a weak hydrogen bond
Journal of Applied Spectroscopy (1984)
-
IR spectra of uranyl complexes with di-n-butylphosphoric acid, tri-n-butylphos phine oxide, and octylamine
Journal of Applied Spectroscopy (1973)
-
Infra-Red Spectra of Some Mono-Esters of Phosphorodichloridous and Phosphonic Acids
Nature (1963)
-
Die Analyse von Pflanzenschutzmittelrückständen
Fresenius' Zeitschrift für analytische Chemie (1962)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.