Abstract
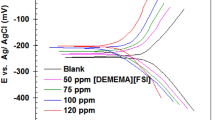
AT each nuclear power station of the Central Electricity Generating Board, the spent-fuel elements will be stored under water for about one hundred days, by which time their radioactivity will have decayed sufficiently to permit them to be removed for reprocessing of the uranium fuel. The water in each cooling pond will be maintained at about pH 11.5 by adding caustic alkali to minimize corrosion of the magnesium alloy can (A12). Although corrosion should be slight, it is still, however, important, since the corrosion product is radioactive due to the presence of active isotopes developed under irradiation from impurities in the magnesium alloy. In addition, accelerated corrosion can be expected on about 50 per cent of the cans in the pond due to carbon deposited on their surface from the carbon dioxide coolant during reactor operation. This type of accelerated corrosion is very sensitive to the presence of chloride ion in the water ; the critical level for rapid attack has been estimated from corrosion tests as less than 10 p.p.m.1. Since all the powar stations are sited near or on the coast, and most have uncovered cooling ponds, the possibility of enhanced corrosion, due to chloride pick-up, is very real, and an electro-chemical assessment of the effect of chloride and carbon on magnesium alloy corrosion is in progress.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Masterson, H. G., and Harrison, J. T., Fuel Element Storage at the Nuclear Power Stations of the Central Electricity Generating Board, First Intern. Congr. Metallic Corrosion, London (1961).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
JONES, D., MASTERSON, H. Effect of Chloride Concentration on the Aqueous Corrosion of a Magnesium Alloy. Nature 191, 165–166 (1961). https://doi.org/10.1038/191165a0
Issue Date:
DOI: https://doi.org/10.1038/191165a0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.