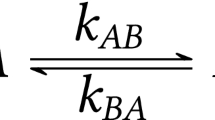Abstract
HIGH-resolution nuclear magnetic resonance spectroscopy affords a uniquely penetrating method of studying co-ordinate complex formation between boron trifluoride and electron donor substances. The examples investigated so far comprise oxygen compounds (water, alcohols, ethers and ketones), but the method promises wide application. Details of a typical experiment follow. Fluorine magnetic resonance (at a frequency of 34.2 Mc./s.) is observed in a quantitatively prepared liquid mixture of two donor substances (as, say, methanol and ethanol) with boron trifluoride, the latter in quantity insufficient to saturate both donors. The temperature is controlled during a given measurement, the usable range extending from somewhat above room temperature to about − 100° C. At sufficiently low temperatures two relatively narrow resonance peaks are seen, separated in the case cited by some 70 c./s. These correspond to the co-ordination complexes of the respective donors. Measurement of the peak areas (assumed to be proportional to the volume concentrations) in mixtures of various composition indicates that reversible equilibrium is established between the two donors and their respective complexes. The mass law constant may thus be measured. The values at a series of different temperatures yield the corresponding enthalpy and standard entropy changes for the interconversion reaction.
Similar content being viewed by others
Article PDF
References
Ogg, R. A., J. Chem. Phys., 22, 560 (1954); 26, 1341 (1957) ; Disc. Farad. Soc., 17, 215 (1954).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DIEHL, P., OGG, R. Nuclear Magnetic Resonance Studies of Boron Trifluoride Complex Formation. Nature 180, 1114 (1957). https://doi.org/10.1038/1801114a0
Issue Date:
DOI: https://doi.org/10.1038/1801114a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.