Abstract
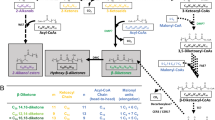
THERE is now considerable evidence to show that the synthesis of isoprenoid compounds in both plants and animals may proceed by the utilization of acetate as the sole primary carbon source1,2, and that the pathways by which the isoprenoid monomer is derived from acetate may have a number of carboxylate intermediates in common, for example, ββ′-dimethyl acrylate and β-hydroxy-β-methyl glutarate1,3. Before taking part in these metabolic reactions acetate first undergoes activation by coupling with coenzyme A, and this form of carboxyl activation is then transmitted through subsequent intermediates arising from acetate. An examination of the latex and tissues of Hevea brasiliensis for such activation systems is briefly reported here.
Similar content being viewed by others
Article PDF
References
Arreguin, B., Bonner, J., and Wood, B. J., Arch. Biochem., 31, 234 (1951).
Cornforth, J. W., and Popják, G., Biochem. J., 58, 403 (1954).
Rudney, H., J. Amer. Chem. Soc., 76, 2595 (1954).
Beinert, H., Green, D. E., Hele, P., Hift, H., Von Korff, R. W., and Ramakrishnan, C. V., J. Biol. Chem., 203, 35 (1953).
Kornberg, A., and Pricer, jun., W. E., J. Biol. Chem., 204, 329 (1953).
Thompson, A. R., Aust. J. Sci. Res., Series B, 4, 180 (1951).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
PATRICK, A. Metabolism of Acetate in Hevea brasiliensis . Nature 180, 37 (1957). https://doi.org/10.1038/180037a0
Issue Date:
DOI: https://doi.org/10.1038/180037a0
This article is cited by
-
Biosynthesis of Rubber
Nature (1959)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.